Zearalenone
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
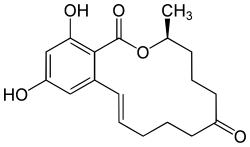
(3S,11E)-14,16-Dihydroxy-3-methyl-3,4,5,6,9,10-hexahydro-1H-2-benzoxacyclotetradecine-1,7(8H)-dione | |
| Other names
Mycotoxin F2
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.038.043 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H22O5 | |
| Molar mass | 318.369 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Zearalenone (ZEN), also known as RAL and F-2 mycotoxin, is a potent estrogenic metabolite produced by some Fusarium and Gibberella species.[1] Specifically, the Gibberella zeae, the fungal species where zearalenone was initially detected, in its asexual/anamorph stage is known as Fusarium graminearum.[2] Several Fusarium species produce toxic substances of considerable concern to livestock and poultry producers, namely deoxynivalenol, T-2 toxin, HT-2 toxin, diacetoxyscirpenol (DAS) and zearalenone. Particularly, ZEN is produced by Fusarium graminearum, Fusarium culmorum, Fusarium cerealis, Fusarium equiseti,[3] Fusarium verticillioides,[4] and Fusarium incarnatum. Zearalenone is the primary toxin that binds to estrogen receptors, causing infertility, abortion or other breeding problems, especially in swine.[4] Often, ZEN is detected together with deoxynivalenol in contaminated samples and its toxicity needs to be considered in combination with the presence of other toxins.[5]
Zearalenone is heat-stable and is found worldwide in a number of cereal crops, such as maize, barley, oats, wheat, rice, and sorghum.[6][7][8] Its production increases when the climate is warm with air humidity at or above twenty percent.[4] The environmental pH plays also a role in the toxin's production. When temperatures fall to 15 °C, alkaline soils still support ZEN production. At the preferred Fusarium temperature, which ranges between 25 °C and 30 °C, neutral pH results in the greatest toxin production.[9]
In addition to its actions on the classical estrogen receptors, zearalenone has been found to act as an agonist of the GPER (GPR30).[8]
Chemical and physical properties
Zearalenone is a white crystalline solid, with molecular formula C18H22O5 and 318.364 g/mol molecular weight. It is a resorcyclic acid lactone. It exhibits blue-green fluorescence when excited by long wavelength ultraviolet (UV) light (360 nm) and a more intense green fluorescence when excited with short wavelength UV light (260 nm).[4] In methanol, UV absorption maxima occur at 236 (e = 29,700), 274 (e = 13,909) and 316 nm (e = 6,020). Maximum fluorescence in ethanol occurs with irradiation at 314 nm and with emission at 450 nm. Solubility in water is about 0.002 g/100 mL. It is slightly soluble in hexane and progressively more so in benzene, acetonitrile, methylene chloride, methanol, ethanol, and acetone. It is also soluble in aqueous alkali.[citation needed]
The naturally occurring isomer trans-zearalenone (trans-ZEN) is transformed by ultraviolet irradiation to cis-zearalenone (cis-ZEN).[10]
Metabolic pathways and products
Zearalenone is metabolically transformed to α-zearalenol (α-Zel) or (α-Zol), β-zearalenol (β-Zel) or (β-Zol), α-zearalanol (α-Zal), β-zearalanol (β-Zal), and zearalanone (ZAN) in animals.[9][11] The relative composition of these metabolic products varies by species. In pigs, cows and ducks, α-Zel is the dominant form detected.[12][13][4] In humans, both α-Zel and β-Zel are seen in urine samples, with the beta form being prevalent.[14] In chickens, β-Zel is the dominant form and in plant cells, the metabolic product zeralenonne-14-O-β-glucoside has been detected.[4] Additionally, in the organs of animals these metabolic products are further modified to yield zearalenone-14-glucuronide (ZEN-14GlcA), α-zearalenol-glucuronide (α-Zel-14G) and β-zearalenol-glucuronide (β-Zel-14G).[15]
Dermal exposure
Zearalenone can permeate through the human skin.[16] However, no significant hormonal effects are expected after dermal contact in normal agricultural or residential environments.
Reproduction
Zearalenone structure is similar to estrogens and α-zearalenol binds with an even greater affinity estrogen receptors, while β-zearalenol's affinity is lower than both the parent compound's and α-Zel's binding affinity.[4] This identifies ZEN and its metabolites as xenoestrogens.[3] The human and livestock exposure to ZEN through the diet poses health concern due to the onset of several sexual disorders and alterations in the development of sexual organs.[17][18] There are reliable case reports of early puberty in girls chronically exposed to ZEN in various regions of the world.[19] In mice, ZEN consumption was linked to a decline of potent sperm and egg cells, an increase to double-stranded breaks in DNA and activation of DNA repair mechanisms, followed by embryonic development challenges that reduced the viability of offspring.[11]
Sampling and analysis
In common with other mycotoxins, sampling food commodities for zearalenone must be carried out to obtain samples representative of the consignment under test. Commonly used extraction solvents are aqueous mixtures of methanol, acetonitrile, or ethyl acetate followed by a range of different clean-up procedures that depend in part on the food and on the detection method in use. Thin-layer chromatography (TLC) methods and high-performance liquid chromatography (HPLC) are commonly used. The TLC method for zearalenone is: normal phase silica gel plates, the eluent: 90% dichloromethane, 10% v/v acetone; or reverse phase C18 silica plates; the eluent: 90% v/v methanol, 10% water. Zearalenone gives unmistakable blue luminiscence under UV.[1] HPLC alone is not sufficient, as it may often yield false positive results. Today, HPLC-MS/MS analysis is used to quantify and confirm the presence of zearalenone.
Typically, the representative sample is commuted and homogenized then few grams are used for extraction with acetonitrile/water mixture. The procedure is the widely used QuEChERS method that quickly and effectively extracts small molecules, like mycotoxins and pesticides, from complex food matrices and animal tissues. The determination step relies on liquid chromatography - mass-spectrometry (LC-MS/MS).[15] Another approach for the analysis of ZEA, without the requirement of expensive instrumentation, is developing specific peptide mimetic with the bioluminescent Gaussia luciferase fused as one protein that can bind specifically to ZEA.[20]
See also
References
- ^ a b "Zearalenone". Fermentek. January 2002.
- ^ Liu J, Applegate T (June 2020). "Zearalenone (ZEN) in Livestock and Poultry: Dose, Toxicokinetics, Toxicity and Estrogenicity". Toxins. 12 (6): 377. doi:10.3390/toxins12060377. PMC 7354539. PMID 32517357.
- ^ a b Bulgaru CV, Marin DE, Pistol GC, Taranu I (March 2021). "Zearalenone and the Immune Response". Toxins. 13 (4): 248. doi:10.3390/toxins13040248. PMC 8066068. PMID 33807171.
- ^ a b c d e f g Ropejko K, Twarużek M (January 2021). "Zearalenone and Its Metabolites-General Overview, Occurrence, and Toxicity". Toxins. 13 (1): 35. doi:10.3390/toxins13010035. PMC 7825134. PMID 33418872.
- ^ Peillod C, Laborde M, Travel A, Mika A, Bailly JD, Cleva D, et al. (February 2021). "Toxic Effects of Fumonisins, Deoxynivalenol and Zearalenone Alone and in Combination in Ducks Fed the Maximum EUTolerated Level". Toxins. 13 (2): 152. doi:10.3390/toxins13020152. PMC 7920068. PMID 33669302.
- ^ Kuiper-Goodman T, Scott PM, Watanabe H (September 1987). "Risk assessment of the mycotoxin zearalenone". Regulatory Toxicology and Pharmacology. 7 (3): 253–306. doi:10.1016/0273-2300(87)90037-7. PMID 2961013.
- ^ Tanaka T, Hasegawa A, Yamamoto S, Lee US, Sugiura Y, Ueno Y (1988). "Worldwide Contamination of Cereals by the Fusarium Mycotoxins Nivalenol, Deoxynivalenol, and Zearalenone. 1. Survey of 19 Countries". Journal of Agricultural and Food Chemistry. 36 (5). American Chemical Society: 979–983. Bibcode:1988JAFC...36..979T. doi:10.1021/jf00083a019.
- ^ a b Prossnitz ER, Barton M (May 2014). "Estrogen biology: new insights into GPER function and clinical opportunities". Molecular and Cellular Endocrinology. 389 (1–2): 71–83. doi:10.1016/j.mce.2014.02.002. PMC 4040308. PMID 24530924.
- ^ a b Mahato DK, Devi S, Pandhi S, Sharma B, Maurya KK, Mishra S, et al. (January 2021). "Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review". Toxins. 13 (2): 92. doi:10.3390/toxins13020092. PMC 7912641. PMID 33530606.
- ^ Brezina U, Kersten S, Valenta H, Sperfeld P, Riedel J, Dänicke S (November 2013). "UV-induced cis-trans isomerization of zearalenone in contaminated maize". Mycotoxin Research. 29 (4): 221–227. doi:10.1007/s12550-013-0178-7. PMID 24018604. S2CID 17466231.
- ^ a b Yang D, Jiang X, Sun J, Li X, Li X, Jiao R, et al. (September 2018). "Toxic effects of zearalenone on gametogenesis and embryonic development: A molecular point of review". Food and Chemical Toxicology. 119: 24–30. doi:10.1016/j.fct.2018.06.003. PMID 29864477. S2CID 46927149.
- ^ Peillod C, Laborde M, Travel A, Mika A, Bailly JD, Cleva D, et al. (February 2021). "Toxic Effects of Fumonisins, Deoxynivalenol and Zearalenone Alone and in Combination in Ducks Fed the Maximum EUTolerated Level". Toxins. 13 (2): 152. doi:10.3390/toxins13020152. PMC 7920068. PMID 33669302.
- ^ Gruber-Dorninger C, Faas J, Doupovec B, Aleschko M, Stoiber C, Höbartner-Gußl A, et al. (January 2021). "Metabolism of Zearalenone in the Rumen of Dairy Cows with and without Application of a Zearalenone-Degrading Enzyme". Toxins. 13 (2): 84. doi:10.3390/toxins13020084. PMC 7911295. PMID 33499402.
- ^ Al-Jaal B, Latiff A, Salama S, Hussain HM, Al-Thani NA, Al-Naimi N, et al. (April 2021). "Analysis of Multiple Mycotoxins in the Qatari Population and Their Relation to Markers of Oxidative Stress". Toxins. 13 (4): 267. doi:10.3390/toxins13040267. PMC 8068385. PMID 33917988.
- ^ a b Yan Z, Wang L, Wang J, Tan Y, Yu D, Chang X, et al. (March 2018). "A QuEChERS-Based Liquid Chromatography-Tandem Mass Spectrometry Method for the Simultaneous Determination of Nine Zearalenone-Like Mycotoxins in Pigs". Toxins. 10 (3): 129. doi:10.3390/toxins10030129. PMC 5869417. PMID 29558416.
- ^ Boonen J, Malysheva SV, Taevernier L, Diana Di Mavungu J, De Saeger S, De Spiegeleer B (November 2012). "Human skin penetration of selected model mycotoxins". Toxicology. 301 (1–3): 21–32. Bibcode:2012Toxgy.301...21B. doi:10.1016/j.tox.2012.06.012. PMID 22749975.
- ^ Massart F, Saggese G (April 2010). "Oestrogenic mycotoxin exposures and precocious pubertal development". International Journal of Andrology. 33 (2): 369–376. doi:10.1111/j.1365-2605.2009.01009.x. PMID 20002219.
- ^ Schoevers EJ, Santos RR, Colenbrander B, Fink-Gremmels J, Roelen BA (August 2012). "Transgenerational toxicity of Zearalenone in pigs". Reproductive Toxicology. 34 (1): 110–119. Bibcode:2012RepTx..34..110S. doi:10.1016/j.reprotox.2012.03.004. PMID 22484360.
- ^ Hueza IM, Raspantini PC, Raspantini LE, Latorre AO, Górniak SL (March 2014). "Zearalenone, an estrogenic mycotoxin, is an immunotoxic compound". Toxins. 6 (3): 1080–1095. doi:10.3390/toxins6031080. PMC 3968378. PMID 24632555.
- ^ Peltomaa R, Fikacek S, Benito-Peña E, Barderas R, Head T, Deo S, et al. (September 2020). "Bioluminescent detection of zearalenone using recombinant peptidomimetic Gaussia luciferase fusion protein". Mikrochimica Acta. 187 (10) 547. doi:10.1007/s00604-020-04538-7. PMC 7938698. PMID 32886242.
External links
 Media related to Zearalenone at Wikimedia Commons
Media related to Zearalenone at Wikimedia Commons- Eriksen GS, Pennington J, Schlatter J (2000). "Zearalenone". WHO International Programme on Chemical Safety - Safety Evaluation of Certain Food Additives and Contaminants. Inchem. WHO Food Additives Series.
Notes
This article is a direct transclusion of the Wikipedia article and therefore may not meet the same editing standards as LIMSwiki.









