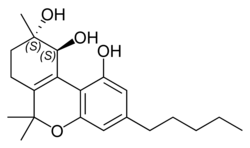
Cannabitriol
From LIMSWiki
Jump to navigationJump to search
 | |
| Clinical data | |
|---|---|
| Other names | (+)-CBT, (S,S)-9,10-Dihydroxy-Δ6a(10a)-THC |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C21H30O4 |
| Molar mass | 346.467 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Cannabitriol (CBT) is a phytocannabinoid first isolated in 1966,[1][2][3] an oxidation product of tetrahydrocannabinol (THC) which has been identified both as a trace component of cannabis and as a metabolite in cannabis users.[4] Its pharmacology has been little studied, though it has been found to act as an antiestrogen and aromatase inhibitor.[5][6]
See also
References
- ^ Obata Y, Ishikawa Y (June 1966). "Studies on the constituents of hemp plant (Cannabis sativa L.) Part III. Isolation of a gibbs-positive compound from Japanese hemp". Agricultural and Biological Chemistry. 30 (6): 619–620. doi:10.1080/00021369.1966.10858651.
- ^ Chan WR, Magnus KE, Watson HA (March 1976). "The structure of cannabitriol". Experientia. 32 (3): 283–4. doi:10.1007/BF01940792. PMID 1253891. S2CID 2679030.
- ^ Elsohly MA, El-Feraly FS, Turner CE (1977). "Isolation and characterization of (+)-cannabitriol and (-)-10-ethoxy-9-hydroxy-delta 6a[10a]-tetrahydrocannabinol: two new cannabinoids from Cannabis sativa L. extract". Lloydia. 40 (3): 275–80. PMID 895385.
- ^ White RM (January 2018). "Instability and poor recovery of cannabinoids in urine, oral fluid, and hair". Forensic Science Review. 30 (1): 33–49. PMID 29273570.
- ^ Baroi S, Saha A, Bachar R, Bachar SC (June 2020). "Cannabinoid as Potential Aromatase Inhibitor through Molecular Modeling and Screening for Anti-Cancer Activity". Dhaka University Journal of Pharmaceutical Sciences. 19 (1): 47–58. doi:10.3329/dujps.v19i1.47818. S2CID 225712476.
- ^ Kikiowo B, Ogunleye AJ, Iwaloye O, Ijatuyi TT, Adelakun NS, Alashe WO (2021). "Induced Fit Docking and Automated QSAR Studies Reveal the ER-α Inhibitory Activity of Cannabis sativa in Breast Cancer". Recent Patents on Anti-Cancer Drug Discovery. 16 (2): 273–284. doi:10.2174/1574892816666210201115359. PMID 33563181. S2CID 231865568.
External links
Notes
This article is a direct transclusion of the Wikipedia article and therefore may not meet the same editing standards as LIMSwiki.









