Journal:AI4Green: An open-source ELN for green and sustainable chemistry
| Full article title | AI4Green: An open-source ELN for green and sustainable chemistry |
|---|---|
| Journal | Journal of Chemical Information and Modeling |
| Author(s) | Boobier, Samuel; Davies, Joseph C.; Derbenev, Ivan N.; Handley, Christopher M.; Hirst, Jonathan D. |
| Author affiliation(s) | University of Nottingham |
| Year published | 2023 |
| Volume and issue | 63(10) |
| Page(s) | 2895–2901 |
| DOI | 10.1021/acs.jcim.3c00306 |
| ISSN | 1549-960X |
| Distribution license | Creative Commons Attribution 4.0 International |
| Website | https://pubs.acs.org/doi/10.1021/acs.jcim.3c00306 |
| Download | https://pubs.acs.org/doi/pdf/10.1021/acs.jcim.3c00306 |
Abstract
This paper presents the free and open-source, web-based electronic laboratory notebook (ELN) AI4Green, which combines features such as data archiving, collaboration tools, and green and sustainability metrics for organic chemistry. AI4Green offers the core functionality of an ELN, namely, the ability to store reactions securely and share them among different members of a research team. As users plan their reactions and record them in the ELN, green and sustainable chemistry is encouraged by automatically calculating green metrics and color-coding hazards, solvents, and reaction conditions. The interface links a database constructed from data extracted from PubChem, enabling the automatic collation of information for reactions. The application’s design facilitates the development of auxiliary sustainability applications, such as our Solvent Guide module. As more reaction data are captured, subsequent work will focus on providing “intelligent” sustainability suggestions to the user.
Keywords: electronic laboratory notebook, ELN, green and sustainable chemistry, open-source, data sharing
Introduction
For researchers to communicate their findings between their team and the wider scientific community, data must be shared and stored. Paper-based laboratory notebooks are traditionally used to record experiments, and little has changed over the last few decades, despite the ubiquity of digital technology. Over the past 20 years, electronic laboratory notebooks (ELNs) have become more prevalent as the benefits of digitization are realized.[1] Despite this, there remains a significant barrier to the uptake of ELNs, especially in the academic community.[2] In 2017, a survey at a BioSistemika webinar revealed that only 7% of respondents used an ELN in their daily laboratory routine.[2] Another survey from the same study showed that the main barriers were the cost associated with implementing an ELN and the system’s usability.
Recently, in a comprehensive comparison of commercial and open-source ELNs, it was discovered that the majority of the 96 currently active ELNs are commercial.[3] It was also noted that open-source codebases have the advantage that users could more directly contribute to the development of new features and have more control over the underlying software. However, there is more onus on the institution to install, host, and maintain the infrastructure. Chemotion is an open-source ELN designed for synthetic chemistry with a growing user base and a strong focus on data sharing and integrity[4][5], but not a particular emphasis on green and sustainable chemistry. Another open-source solution is eLabFTW, an ELN suitable for storing data from various scientific disciplines.[6]
Research data management is fundamental to scientific research.[7] Transitioning from paper-based laboratory notebooks to ELNs is crucial for adhering to data standards when reporting and publishing studies.[8] ELNs are also vital in making data FAIR (findable, accessible, interoperable, and reusable).[9] ELNs allow data sharing among colleagues and institutions, while also facilitating public access.[10] Open science[11][12] can also be enabled using ELNs, where data is curated using a standard data format, expediting data searches and preparation for machine learning (ML), where large data sets are often required to train insightful models. Recent examples of such databases include the Open Reaction Database[13] and the Chemotion Repository.[14]
Sustainability and reducing waste are vital considerations in laboratory-based projects. "Sustainable" refers to both the environmental and socio-economic impacts of a process.[15] Making processes more sustainable is not just a requirement of government regulations. There are also the benefits of cost reductions, improved worker health and safety, and the reduction of impact on the environment.[16][17] Current software tools for green and sustainable chemistry have recently been reviewed.[18] ELNs also offer the opportunity for collecting data that can be used to monitor sustainability targets (such as the reduction of hazardous solvents) and share knowledge among colleagues.[19]
In this work, we present AI4Green, designed to fulfill the core functionality of an ELN for synthetic organic chemistry in academic and industry settings, while also encouraging green and sustainable chemistry. The software automatically presents the hazards and sustainability of an inputted reaction by calculating sustainability metrics and a color-coded assessment of solvents and reaction conditions. While the web application is open-source, the software is provided in a manner that has a low barrier to installation and hosting, has a user-friendly interface, and is easily customizable. As the number of users grows, the captured reaction data will be subsequently leveraged using ML to provide “intelligent” suggestions to users on improving their reactions’ sustainability.
Implementation
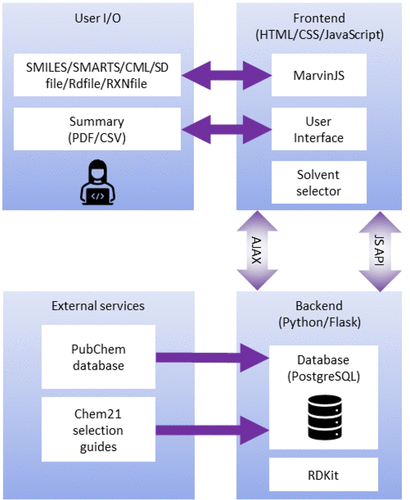
AI4Green is a web application written in Python, JavaScript, HTML, and CSS (Figure 1). The application is hosted on the cloud and available for general use at https://ai4green.app. Alternatively, visit the GitHub page for simple instructions detailing installation and hosting via Docker either locally or on an organization’s local server.
|
The backend server is built with Python and Flask and is linked to a Postgres relational database. The advantage of using Python for the backend is the access to many popular standard chemistry libraries, such as RDKit[20], and a low barrier for developers to add additional features. Flask is a well-documented and easy-to-use web application framework[21], and blueprints provide a clear structure to the code and facilitate expansion. Flask separates the Python backend from the JavaScript, HTML, and CSS-controlled frontend, where users input their data via Marvin JS[22] chemical editor or in a number of formats, including Simplified Molecular-Input Line-Entry System (SMILES).[23] JavaScript AJAX requests are used to update pages dynamically, e.g., automatically calculating green and sustainable metrics when inputting user data. A summary and sustainability report are presented back to the user which can be exported as a PDF or CSV file. A single database was implemented in Postgres, constructed from compound data extracted from PubChem[24] and CHEM21 sustainability data[25], to provide chemical information automatically, and separated into tables for users, workgroups, workbooks, solvents, hazards, compounds (reagents), and reactions.
Results
Workgroups, workbooks, and user types
AI4Green installations may have one or more admin users. These users, typically system administrators for an institution, review requests to make new workgroups and can monitor the number of users, compounds, and reactions on the server. Users must register for an account to use AI4Green, at which point those in the principal investigator (PI) or equivalent roles, are prompted to make a workgroup as a space for their research group; other users are directed to join the workgroup their PI has created.
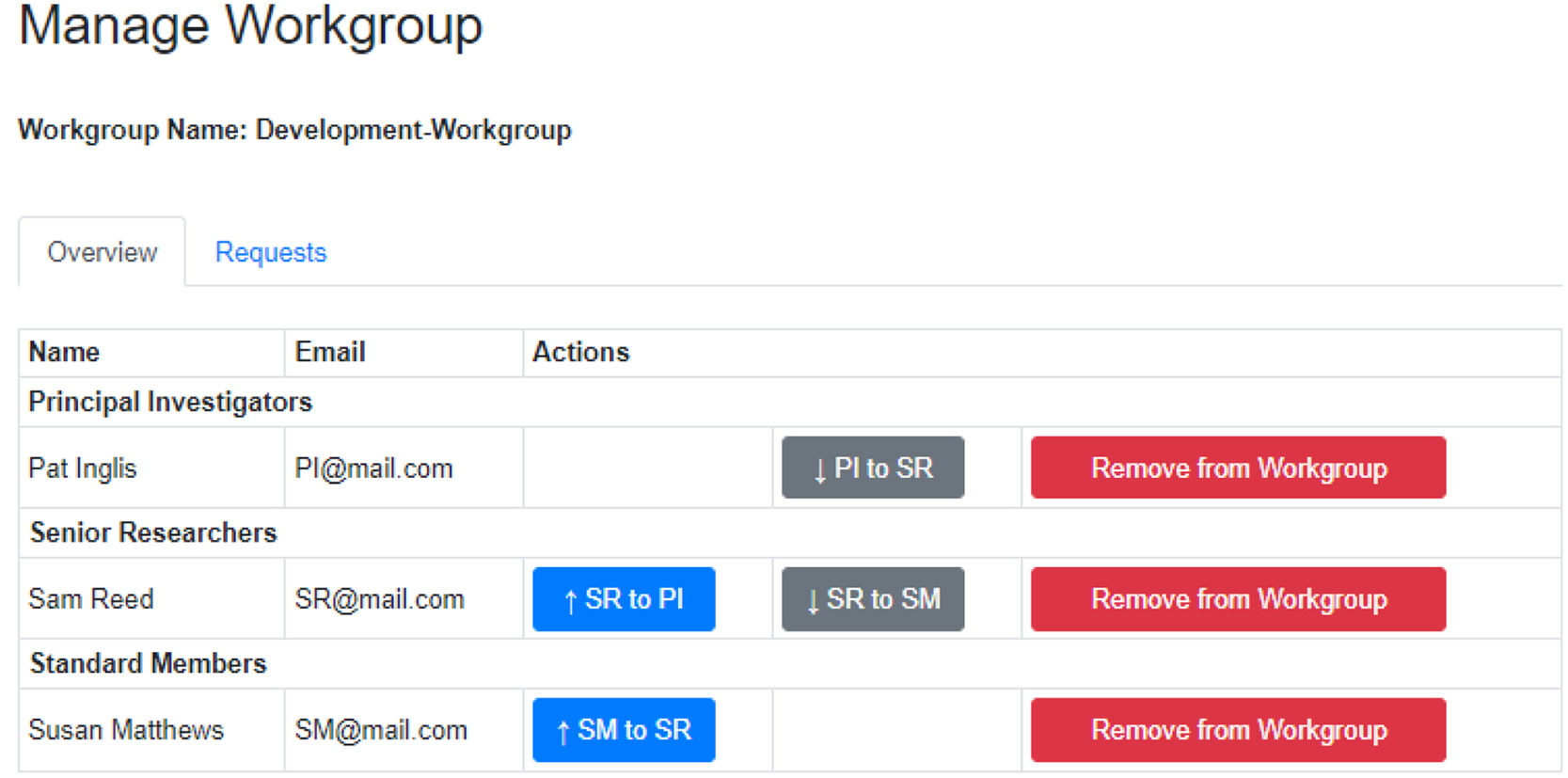
Within workgroups, there are workbooks that are designed to contain reactions for a specific project (Figure 2). Workgroups have three roles with different permission levels. PIs are the workgroup owner and have full permission to create workbooks and add or remove users from the workgroup and any workbooks within it. It is permissible to have two or more PIs in a workgroup. The senior researcher role, suitable for postdoctoral researchers or equivalent, can create new workbooks and add or remove users to these workbooks. They have no such rights for workgroups. The standard member role, suitable for postgraduate researchers or equivalent, have no editing rights but can request to be added to workbooks.
Using this flexible approach, a user can belong to multiple workgroups in different roles, e.g., a PI in one workgroup and a senior researcher in another. Reactions are only shared within the same workbook, as are any novel compounds added to the database. A user owns the reactions they create, which are also available as read-only entries to all members of the workbook, thus enabling data sharing between team members. This is especially useful when teams are spread over multiple locations while preserving data privacy.
|
Reaction Builder
The core functionality of AI4Green is the Reaction Builder module. In later sections, we give further details on the different components of the Reaction Builder. Reactions can be created by navigating to a workgroup, choosing a workbook, and selecting “New Reaction.” The user is prompted to enter a name for the reaction, which must be unique within the workbook. In addition, a unique code is also assigned to every new reaction. Users draw their reaction into the Marvin JS reaction sketcher. Next, the user is prompted to fill in the Reaction Table, for example, inputting the amount of each reaction component. At this stage, further solvents and reagents can be added. Finally, the Summary Table, which contains several automatically calculated green and sustainable metrics and detailed health and safety information, is generated. The reaction is automatically saved when new changes are made and can be reloaded and edited at a later date. The Summary Table can be exported to PFG or printed for use as a risk assessment.
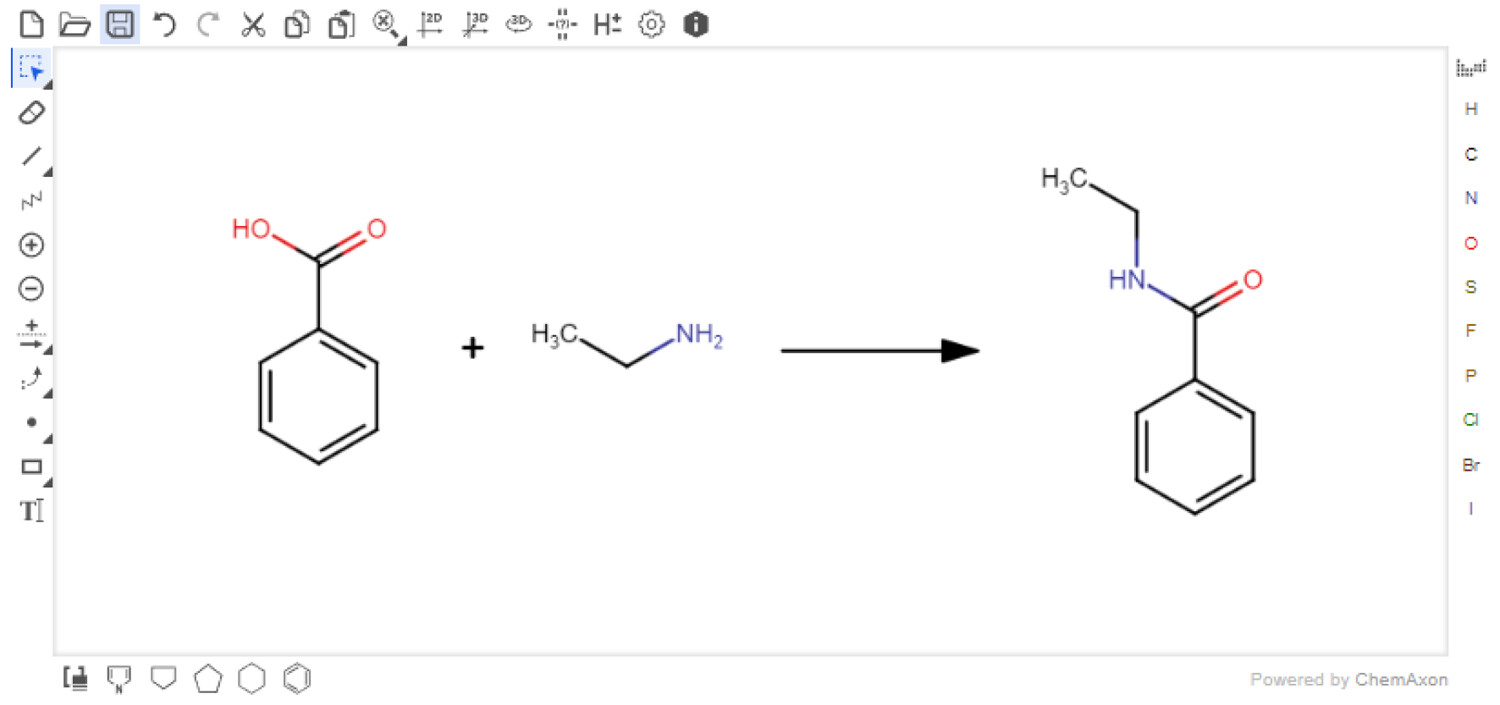
Marvin JS
Users must first input their reaction with the Marvin JS reaction sketcher (Figure 3). This sketcher is easy-to-use and well-documented.[22] For users familiar with other sketchers, it is possible to import structures in several formats, e.g., SMILES[23], which are easily exported from other sketchers. Reagents or solvents above or below the arrow are not currently accepted. However, these can be added directly to the Reaction Table. When the reaction is submitted, the Reaction SMILES (RXSMILES) is exported from the sketcher to the Reaction Table. The database, containing information from PubChem, is queried for the reactants and products in the RXSMILES to obtain density, molecular weight, and hazard codes automatically. All compound data have been collected from PubChem laboratory chemical safety sheets (LCSS). The hazard data are presented as Globally Harmonized System of Classification and Labelling of Chemicals (GHS) hazard codes.[26] The hazard data are only collected from the references provided by the European Chemicals Agency (ECHA).[27]
|
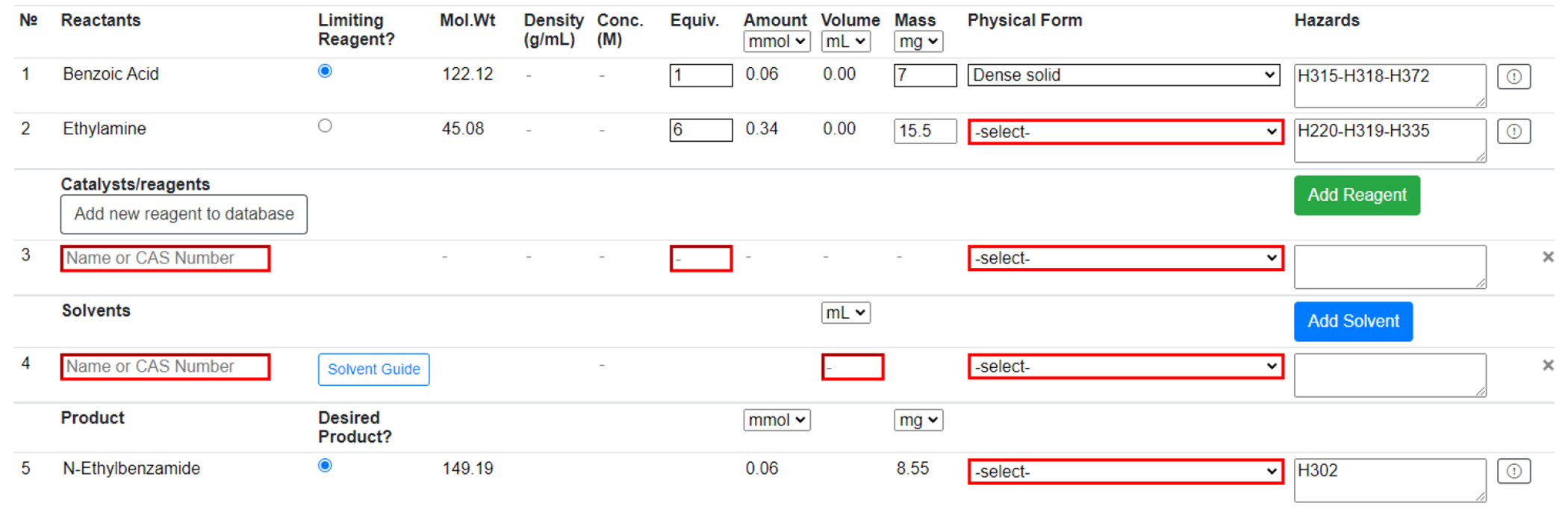
Reaction Table
The user is prompted to populate the Reaction Table (Figure 4) and provide extra information if any reactant or product is not in the database. This “novel compound” is saved to the database and can be reused, but only within the same workbook. Reagents can be added from the PubChem compound database by searching name or Chemical Abstracts Service (CAS) identifier; they can also be added to the database like a novel compound. Solvents can also be added from a predefined list and by searching name or CAS. “Novel solvents” can be added in the same way as “novel compounds”. Solvents are color-coded according to the four-tier CHEM21 classification—recommended, problematic, hazardous, and highly hazardous[25]—providing immediate feedback to the user on the sustainability of their solvent choice. Users will then input the details of their reaction into the Reaction Table. Physical forms of all reactants, reagents, solvents, and products must be provided to assess the reaction’s risk. Additionally, limiting reagent mass and the equivalence of all other reactants and reagents are required to proceed. Any suspected incorrect data from the database can be reported to system administrators for review at any point in the procedure. There is also space to describe the experimental procedure and any observations made during the reaction.
|
Summary Table
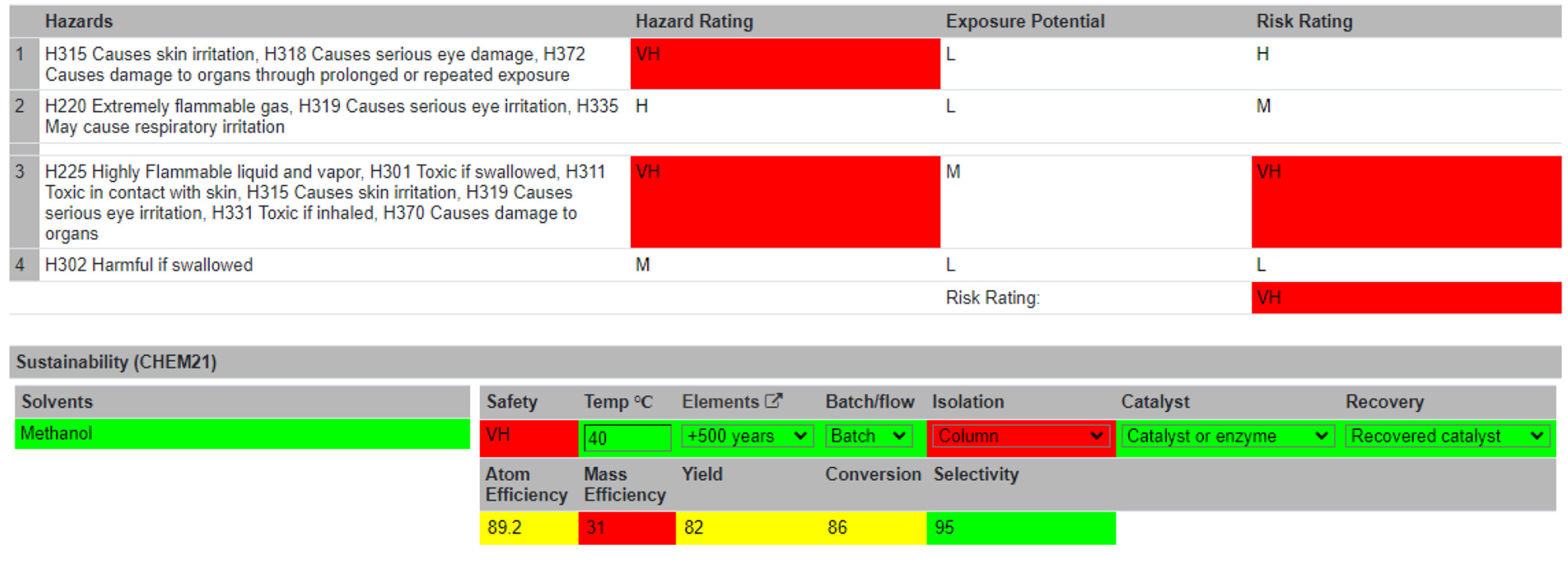
With the Reaction Table complete, users are directed to the Summary Table (Figure 5). Information is automatically passed from the Reaction Table to the Summary Table. Visual assessments of the greenness and sustainability of the reaction are displayed to the users. These are either flagged as unsustainable (red) or given a traffic light system (i.e., red = not recommended/hazardous; yellow = problematic; and green = recommended). The specific colors and shades for these ratings can be altered on the accessibility page. An overall hazard rating is generated from the hazard codes, denoted as Low (L), Medium (M), Hazardous (H), or Very Hazardous (VH). The threshold of the sustainability levels of the following metrics was in accordance with the CHEM21 project.[28] Several of these metrics are calculated automatically, like the sustainability of the chemical elements used in the reaction and the atom efficiency. Other metrics must be inputted by the user, such as the temperature of the reaction, batch or flow reaction conditions, the isolation method, the use of a catalyst, and whether that catalyst was recovered. A risk assessment section follows, which allows users to identify standard protocols, disposal of waste materials, spillage procedures, and any other risks associated with the reaction. An overall risk score can then be computed by self-assessment of the reaction’s hazards, risks, and consequences. Typically, a reaction would be performed at this point. After the reaction run, the user can return to the Summary Table and input unreacted and actual product mass. Using these inputs, four more metrics are computed: mass efficiency, yield, conversion, and selectivity. The reaction can be marked as complete and locked to further editing at this stage. For increased data integrity, reactions modifications are time-stamped in the database. Reactions may currently be searched alphabetically or by most recently created.
|
Exporting data
A reaction can be exported as a PDF, as some institutions may require reaction information and risk assessments to be displayed or filed. It is critical that data can be safely exported from an ELN for the longevity of that data, to move the data to a new system, and for more detailed analysis. The reactions from a workbook can be exported as a single PDF file, multiple PDF files, or a CSV file. The versatility of CSV files, which are machine-readable and can be opened in programs such as Microsoft Excel, makes this file format the preferred option for data offloading.
Sustainability add-ons
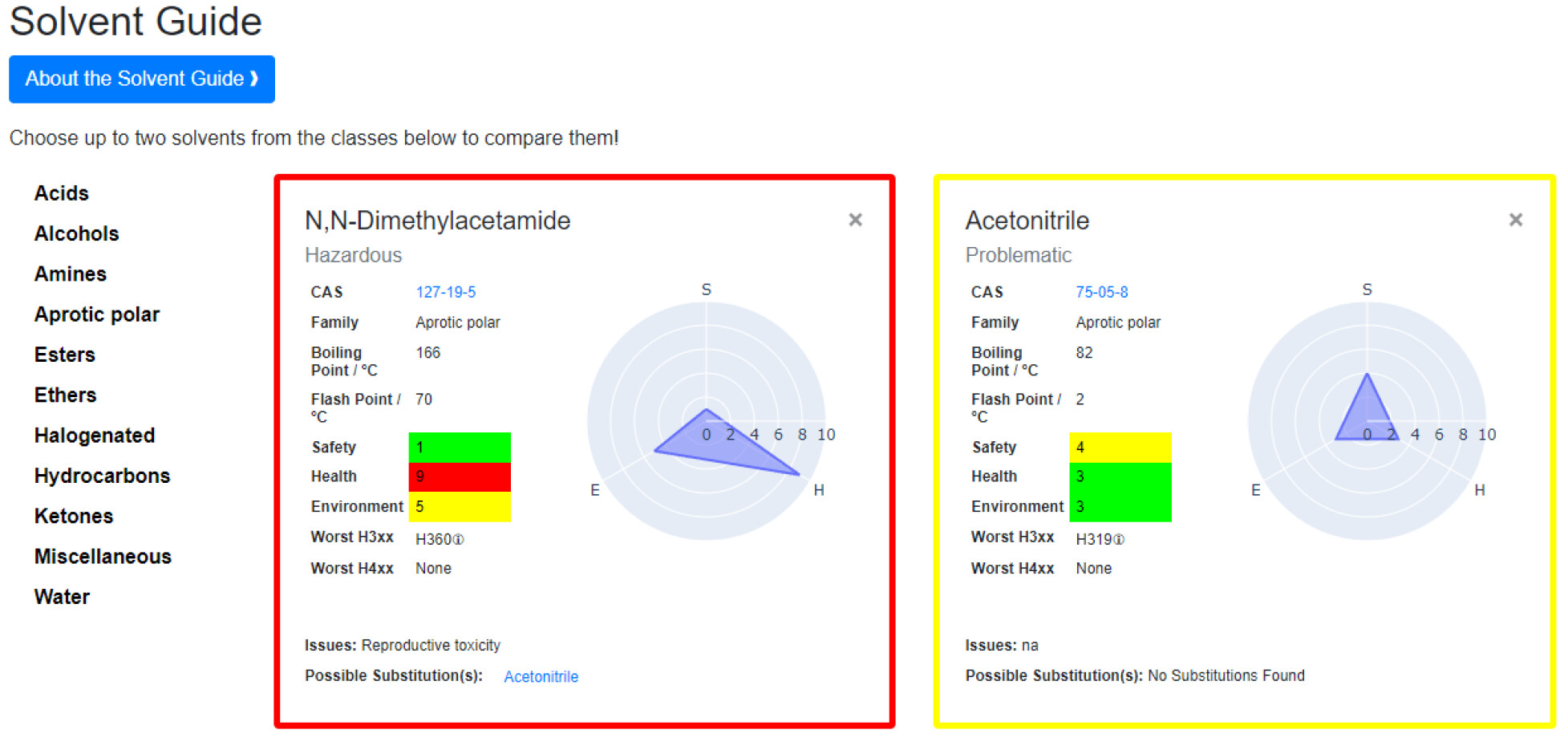
AI4Green is open-source, and adding new sustainable auxiliary applications is simple. The Python Flask backend, built around blueprints, gives new developers a low barrier. An example of an add-on is the Solvent Guide module (Figure 6), which is a series of solvent flashcards. This can be accessed directly from the top navigation banner or as a user intervention while building a reaction. As previously discussed, the CHEM21 sustainability rating is displayed to the user when a solvent is selected. At this point, the user can open the solvent guide with this preloaded solvent flashcard. A second solvent can be selected for a side-by-side comparison. These solvent flashcards were created using data from the CHEM21 project, where the overall sustainability of the solvent is shown using the four-tier system. This is accompanied by a breakdown of the solvent’s health, safety, and environment scores, with each category given a score out of 10 and a corresponding hazard color. The full methodology for calculating these scores is described in the CHEM21 publication.[25] The CAS number, linked to the PubChem entry for the solvent, is displayed to allow the user to access more information about the solvent. The family of the solvent, boiling point, flash point, and worst hazards are also displayed to guide the user further. For some solvents, a possible substitution is suggested based on an industrial solvent replacement guide and the reason for this substitution.[29] The solvent guide aims to bring the most relevant information to the user on a single page, allowing the solvents to be easily compared side-by-side. This can empower the user to choose a more sustainable and suitable solvent for their reaction at the planning stage. Future add-on applications could include a solvent map for solvent substitution, solvent or reaction conditions prediction for a specific reaction, life cycle analysis of reactions, and retrosynthesis for a target product.
|
User feedback and application development
AI4Green has a growing userbase and is currently used by several academic groups, primarily at the University of Nottingham. As the application has evolved, users have been able to give direct input into new features either by contacting the team directly or by attending our regular user group meetings. Early feedback identified that the ELN must be easy-to-use, convenient for the laboratory chemist, and allow data to be easily shared throughout the group. Specific examples of features added because of user feedback include an automatic unique numbering system for reactions on their creation since users identified that, in a busy lab schedule, several reactions may be worked on concurrently; an autosave feature to ensure data were not accidentally lost if the browser tab is closed without saving; and adding reagents and solvents by CAS number, since this is a common identifier for laboratory chemists.
Conclusion
In this work we present an open-source web application, AI4Green, which combines the practical benefits of an ELN alongside a framework for encouraging green and sustainable chemistry. Despite the manifold benefits, many academic groups are yet to adopt usage of an ELN. AI4Green provides an accessible platform for chemists to store and share their research and receive feedback on the sustainability of their reactions. Research teams can be easily organized into workgroups and workbooks, with different levels of permission for different user types. The Reaction Builder provides a semi-automated route to generating sustainability, hazard reports, and risk assessments. The Solvent Guide is an example of an add-on application to encourage sustainable solvent selection.
There are, however, still many features to implement in AI4Green. Data sharing between different workgroups or to the public is not yet possible. We also aim to use inputted reaction data to make intelligent sustainability suggestions. This may be to suggest using a less hazardous solvent or reagent, predict milder suitable reaction conditions, or simulate life cycle analysis for process scale-up. There will be challenges in preserving data privacy for users, who will be able to decide whether to share data fully, partially, or not at all. Additional features to be implemented include searching reactions by component name or a substructure, a mechanism for PIs to approve reactions, and a sustainability dashboard to allow groups to set sustainability goals and track their progress toward them.
AI4Green provides an exciting initial framework to unite an ELN with sustainable chemistry. It has a growing user base and rapidly evolving functionality.
Supporting information
Abbreviations, acronyms, and initialisms
- CAS: Chemical Abstracts Service
- ECHA: European Chemicals Agency
- ELN: electronic laboratory notebook
- GHS: Globally Harmonized System of Classification and Labelling of Chemicals
- LCSS: laboratory chemical safety sheets
- PI: principal investigator
- RXSMILE: Reaction SMILES
- SMILES: Simplified Molecular-Input Line-Entry System
Acknowledgements
We thank all the users of AI4Green for their valuable feedback and Aimie Garces, James Dowden, Jamie Twycross, and Zakariyyaa Siddiq for many useful discussions.
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. S.B., J.C.D., C.M.H., and I.N.D. contributed to the development of the software. J.D.H. conceived and supervised the project.
Funding
This work and J.D.H. are supported by the Royal Academy of Engineering under the Chairs in Emerging Technologies scheme [CiET2021_17]. The work was also supported in part by the Engineering and Physical Sciences Research Council (EPSRC) [grant number EP/S035990/1].
Data availability
AI4Green is open-source and released under the AGPL-3.0 license. Full source code, installation instructions, and links to our video tutorials and user guides can be found at GitHub.
Conflict of interest
The authors declare no competing financial interest.
References
- ↑ Bird, Colin L.; Willoughby, Cerys; Frey, Jeremy G. (2013). "Laboratory notebooks in the digital era: the role of ELNs in record keeping for chemistry and other sciences" (in en). Chemical Society Reviews 42 (20): 8157. doi:10.1039/c3cs60122f. ISSN 0306-0012. http://xlink.rsc.org/?DOI=c3cs60122f.
- ↑ 2.0 2.1 Kanza, Samantha; Willoughby, Cerys; Gibbins, Nicholas; Whitby, Richard; Frey, Jeremy Graham; Erjavec, Jana; Zupančič, Klemen; Hren, Matjaž et al. (1 December 2017). "Electronic lab notebooks: can they replace paper?" (in en). Journal of Cheminformatics 9 (1): 31. doi:10.1186/s13321-017-0221-3. ISSN 1758-2946. PMC PMC5443717. PMID 29086051. https://jcheminf.biomedcentral.com/articles/10.1186/s13321-017-0221-3.
- ↑ Higgins, Stuart G.; Nogiwa-Valdez, Akemi A.; Stevens, Molly M. (1 February 2022). "Considerations for implementing electronic laboratory notebooks in an academic research environment" (in en). Nature Protocols 17 (2): 179–189. doi:10.1038/s41596-021-00645-8. ISSN 1754-2189. https://www.nature.com/articles/s41596-021-00645-8.
- ↑ Tremouilhac, Pierre; Nguyen, An; Huang, Yu-Chieh; Kotov, Serhii; Lütjohann, Dominic Sebastian; Hübsch, Florian; Jung, Nicole; Bräse, Stefan (1 December 2017). "Chemotion ELN: an Open Source electronic lab notebook for chemists in academia" (in en). Journal of Cheminformatics 9 (1): 54. doi:10.1186/s13321-017-0240-0. ISSN 1758-2946. PMC PMC5612905. PMID 29086216. https://jcheminf.biomedcentral.com/articles/10.1186/s13321-017-0240-0.
- ↑ Kotov, Serhii; Tremouilhac, Pierre; Jung, Nicole; Bräse, Stefan (1 December 2018). "Chemotion-ELN part 2: adaption of an embedded Ketcher editor to advanced research applications" (in en). Journal of Cheminformatics 10 (1): 38. doi:10.1186/s13321-018-0292-9. ISSN 1758-2946. PMC PMC6089857. PMID 30105533. https://jcheminf.biomedcentral.com/articles/10.1186/s13321-018-0292-9.
- ↑ CARPi, Nicolas; Minges, Alexander; Piel, Matthieu (14 April 2017). "eLabFTW: An open source laboratory notebook for research labs". The Journal of Open Source Software 2 (12): 146. doi:10.21105/joss.00146. ISSN 2475-9066. http://joss.theoj.org/papers/10.21105/joss.00146.
- ↑ Kanza, Samantha; Knight, Nicola J. (1 December 2022). "Behind every great research project is great data management" (in en). BMC Research Notes 15 (1): 20. doi:10.1186/s13104-022-05908-5. ISSN 1756-0500. PMC PMC8781028. PMID 35063017. https://bmcresnotes.biomedcentral.com/articles/10.1186/s13104-022-05908-5.
- ↑ Herres‐Pawlis, Sonja; Bach, Felix; Bruno, Ian J.; Chalk, Stuart J.; Jung, Nicole; Liermann, Johannes C.; McEwen, Leah R.; Neumann, Steffen et al. (19 December 2022). "Minimum Information Standards in Chemistry: A Call for Better Research Data Management Practices" (in en). Angewandte Chemie International Edition 61 (51). doi:10.1002/anie.202203038. ISSN 1433-7851. https://onlinelibrary.wiley.com/doi/10.1002/anie.202203038.
- ↑ Wilkinson, Mark D.; Dumontier, Michel; Aalbersberg, IJsbrand Jan; Appleton, Gabrielle; Axton, Myles; Baak, Arie; Blomberg, Niklas; Boiten, Jan-Willem et al. (15 March 2016). "The FAIR Guiding Principles for scientific data management and stewardship" (in en). Scientific Data 3 (1): 160018. doi:10.1038/sdata.2016.18. ISSN 2052-4463. PMC PMC4792175. PMID 26978244. https://www.nature.com/articles/sdata201618.
- ↑ Fink, Fabian; Hüppe, Henrika M.; Jung, Nicole; Hoffmann, Alexander; Herres‐Pawlis, Sonja (1 October 2022). "Sharing is Caring: Guidelines for Sharing in the Electronic Laboratory Notebook (ELN) Chemotion as applied by a Synthesis‐oriented Working Group**" (in en). Chemistry–Methods 2 (10). doi:10.1002/cmtd.202200026. ISSN 2628-9725. https://onlinelibrary.wiley.com/doi/10.1002/cmtd.202200026.
- ↑ Jablonka, Kevin Maik; Patiny, Luc; Smit, Berend (1 April 2022). "Making the collective knowledge of chemistry open and machine actionable" (in en). Nature Chemistry 14 (4): 365–376. doi:10.1038/s41557-022-00910-7. ISSN 1755-4330. https://www.nature.com/articles/s41557-022-00910-7.
- ↑ Baldi, Pierre (9 May 2022). "Call for a Public Open Database of All Chemical Reactions" (in en). Journal of Chemical Information and Modeling 62 (9): 2011–2014. doi:10.1021/acs.jcim.1c01140. ISSN 1549-9596. https://pubs.acs.org/doi/10.1021/acs.jcim.1c01140.
- ↑ Kearnes, Steven M.; Maser, Michael R.; Wleklinski, Michael; Kast, Anton; Doyle, Abigail G.; Dreher, Spencer D.; Hawkins, Joel M.; Jensen, Klavs F. et al. (17 November 2021). "The Open Reaction Database" (in en). Journal of the American Chemical Society 143 (45): 18820–18826. doi:10.1021/jacs.1c09820. ISSN 0002-7863. https://pubs.acs.org/doi/10.1021/jacs.1c09820.
- ↑ Tremouilhac, Pierre; Huang, Pei‐Chi; Lin, Chia‐Lin; Huang, Yu‐Chieh; Nguyen, An; Jung, Nicole; Bach, Felix; Bräse, Stefan (1 January 2021). "Chemotion Repository, a Curated Repository for Reaction Information and Analytical Data" (in en). Chemistry–Methods 1 (1): 8–11. doi:10.1002/cmtd.202000034. ISSN 2628-9725. https://onlinelibrary.wiley.com/doi/10.1002/cmtd.202000034.
- ↑ Horváth, István T. (24 January 2018). "Introduction: Sustainable Chemistry" (in en). Chemical Reviews 118 (2): 369–371. doi:10.1021/acs.chemrev.7b00721. ISSN 0009-2665. https://pubs.acs.org/doi/10.1021/acs.chemrev.7b00721.
- ↑ Schulte, Paul A; McKernan, Lauralynn T; Heidel, Donna S; Okun, Andrea H; Dotson, Gary Scott; Lentz, Thomas J; Geraci, Charles L; Heckel, Pamela E et al. (1 December 2013). "Occupational safety and health, green chemistry, and sustainability: a review of areas of convergence" (in en). Environmental Health 12 (1): 31. doi:10.1186/1476-069X-12-31. ISSN 1476-069X. PMC PMC3639149. PMID 23587312. http://ehjournal.biomedcentral.com/articles/10.1186/1476-069X-12-31.
- ↑ Anastas, Paul T.; Zimmerman, Julie B. (1 October 2018). "The United Nations sustainability goals: How can sustainable chemistry contribute?" (in en). Current Opinion in Green and Sustainable Chemistry 13: 150–153. doi:10.1016/j.cogsc.2018.04.017. https://linkinghub.elsevier.com/retrieve/pii/S245222361830021X.
- ↑ Derbenev, Ivan N.; Dowden, James; Twycross, Jamie; Hirst, Jonathan D. (1 June 2022). "Software tools for green and sustainable chemistry" (in en). Current Opinion in Green and Sustainable Chemistry 35: 100623. doi:10.1016/j.cogsc.2022.100623. https://linkinghub.elsevier.com/retrieve/pii/S2452223622000359.
- ↑ Fantke, Peter; Cinquemani, Claudio; Yaseneva, Polina; De Mello, Jonathas; Schwabe, Henning; Ebeling, Bjoern; Lapkin, Alexei A. (1 November 2021). "Transition to sustainable chemistry through digitalization" (in en). Chem 7 (11): 2866–2882. doi:10.1016/j.chempr.2021.09.012. https://linkinghub.elsevier.com/retrieve/pii/S2451929421004745.
- ↑ "RDKit: Open-Source Cheminformatics Software". T5 Informatics GmbH. https://www.rdkit.org/. Retrieved 27 January 2023.
- ↑ "Flask". Pallets. https://flask.palletsprojects.com/en/2.3.x/. Retrieved 27 January 2023.
- ↑ 22.0 22.1 "Marvin JS User's Guide". Chemaxon. https://docs.chemaxon.com/display/docs/marvin-js-user-s-guide.md. Retrieved 27 January 2023.
- ↑ 23.0 23.1 Weininger, David (1 February 1988). "SMILES, a chemical language and information system. 1. Introduction to methodology and encoding rules" (in en). Journal of Chemical Information and Computer Sciences 28 (1): 31–36. doi:10.1021/ci00057a005. ISSN 0095-2338. https://pubs.acs.org/doi/abs/10.1021/ci00057a005.
- ↑ "PubChem". National Library of Medicine. https://pubchem.ncbi.nlm.nih.gov/. Retrieved 27 January 2023.
- ↑ 25.0 25.1 25.2 Prat, Denis; Wells, Andy; Hayler, John; Sneddon, Helen; McElroy, C. Robert; Abou-Shehada, Sarah; Dunn, Peter J. (2016). "CHEM21 selection guide of classical- and less classical-solvents" (in en). Green Chemistry 18 (1): 288–296. doi:10.1039/C5GC01008J. ISSN 1463-9262. http://xlink.rsc.org/?DOI=C5GC01008J.
- ↑ "About the GHS". United Nations Economic Commission for Europe. 2021. https://unece.org/about-ghs.
- ↑ "Information on Chemicals". European Chemicals Agency. https://echa.europa.eu/information-on-chemicals. Retrieved 27 January 2023.
- ↑ McElroy, C. Robert; Constantinou, Andri; Jones, Leonie C.; Summerton, Louise; Clark, James H. (2015). "Towards a holistic approach to metrics for the 21st century pharmaceutical industry" (in en). Green Chemistry 17 (5): 3111–3121. doi:10.1039/C5GC00340G. ISSN 1463-9262. http://xlink.rsc.org/?DOI=C5GC00340G.
- ↑ Alfonsi, Kim; Colberg, Juan; Dunn, Peter J.; Fevig, Thomas; Jennings, Sandra; Johnson, Timothy A.; Kleine, H. Peter; Knight, Craig et al. (2008). "Green chemistry tools to influence a medicinal chemistry and research chemistry based organisation" (in en). Green Chem. 10 (1): 31–36. doi:10.1039/B711717E. ISSN 1463-9262. http://xlink.rsc.org/?DOI=B711717E.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. Grammar was cleaned up for smoother reading. In some cases important information was missing from the references, and that information was added. This version adds a reference to the GHS.