Journal:Recommended versus certified repositories: Mind the gap
| Full article title | Recommended versus certified repositories: Mind the gap |
|---|---|
| Journal | Data Science Journal |
| Author(s) | Husen, Sean Edward; de Wilde, Zoë G.; de Waard, Anita; Cousijn, Helena |
| Author affiliation(s) | Leiden University, Elsevier |
| Primary contact | Email: s dot e dot husen at hum dot leidenuniv dot nl |
| Year published | 2017 |
| Volume and issue | 16(1) |
| Page(s) | 42 |
| DOI | 10.5334/dsj-2017-042 |
| ISSN | 1683-1470 |
| Distribution license | Creative Commons Attribution 4.0 International |
| Website | https://datascience.codata.org/article/10.5334/dsj-2017-042/ |
| Download | https://datascience.codata.org/articles/10.5334/dsj-2017-042/galley/710/download/ (PDF) |
|
|
This article should not be considered complete until this message box has been removed. This is a work in progress. |
Abstract
Researchers are increasingly required to make research data publicly available in data repositories. Although several organizations propose criteria to recommend and evaluate the quality of data repositories, there is no consensus of what constitutes a good data repository. In this paper, we investigate, first, which data repositories are recommended by various stakeholders (publishers, funders, and community organizations) and second, which repositories are certified by a number of organizations. We then compare these two lists of repositories, and the criteria for recommendation and certification. We find that criteria used by organizations recommending and certifying repositories are similar, although the certification criteria are generally more detailed. We distill the lists of criteria into seven main categories: “Mission,” “Community/Recognition,” “Legal and Contractual Compliance,” “Access/Accessibility,” “Technical Structure/Interface,” “Retrievability,” and “Preservation.” Although the criteria are similar, the lists of repositories that are recommended by the various agencies are very different. Out of all of the recommended repositories, less than six percent obtained certification. As certification is becoming more important, steps should be taken to decrease this gap between recommended and certified repositories, and ensure that certification standards become applicable, and applied, to the repositories which researchers are currently using.
Keywords: data repositories, data management, certification, data quality, data fitness
Introduction
Data sharing and data management are topics that are becoming increasingly important. More information is appearing about their benefits, such as increased citation rates for research papers with associated shared datasets.[1][2] A growing number of funding bodies such as the NIH and the Wellcome Trust[3][4], as well as several journals[5], have installed policies that require research data to be shared.[6] To be able to share data, both now and in the future, datasets not only need to be preserved but also need to be comprehensible and usable for others. To ensure these qualities, research data needs to be managed[7], and data repositories can play a role in maintaining the data in a usable structure.[8] However, using a data repository does not guarantee that the data is usable, since not every repository uses the same procedures and quality metrics, such as applying proper metadata tags.[9] As many repositories have not yet adopted generally accepted standards, it can be difficult for researchers to choose the right repository for their dataset.[7]
Several organizations, including funding agencies, academic publishers, and data organizations provide researchers with lists of supported or recommended repositories, e.g., BioSharing.[10] These lists vary in length, in the number and type of repositories they list, and in their selection criteria for recommendation. In addition, recommendations for data and data sharing are emerging, such as the FAIR Data Principles, guidelines to establish a common ground for all data to be findable, accessible, interoperable, and reusable.[11] Some data repositories are beginning to incorporate the FAIR principles into their policies, such as the UK Data Service[12] and several funders such as the EU Horizon 2020 program and the NIH.[13][14] Lists of recommended repositories and guidelines such as these can help researchers decide how and where to store and share their data.
Next to lists of recommended repositories, there are a number of schemes which specifically certify the quality of data repositories. One of the first of these certification schemes is the Data Seal of Approval (DSA), with an objective "to safeguard data, to ensure high quality and to guide reliable management of data for the future without requiring the implementation of new standards, regulations or high costs."[15] Building upon the DSA certification, but with more elaborate and detailed guidelines[16], is the Network of Expertise in Long-Term Storage of Digital Resources (NESTOR) and the ISO 16363 standard/Trusted Data Repository (TDR). DSA, NESTOR, and TDR form a three-step framework for data repository certification.[16] The ICSU-WDS membership incorporates guidelines from DSA, NESTOR and Trustworthy Repositories Audit & Certification (TRAC), among others, for its data repository framework.[17] Furthermore, the TRAC guidelines were used as a basis for the ISO 16363/TDR guidelines.[18]
Given the multitude of recommendations and certification schemes, we set out to map the current landscape to compare criteria and analyze which repositories are recommended and certified by different parties. This paper is structured as follows: first, we investigate which repositories have been recommended and certified by different organizations. Next, we provide an analysis of the criteria used by organizations recommending repositories and the criteria used by certification schemes, and then derive a set of shared criteria for recommendation and certification. Lastly, we explore what this tells us about the overlap between recommendations and certifications.
Methods
Lists of repositories
Recommended repositories
To examine which repositories are being recommended, we looked at the recommendations of 17 different organizations, including academic publishers, funding agencies, and data organizations. These lists of recommended repositories include all the available recommendation lists currently found on the BioSharing (now "FAIRsharing") website under the Recommendations tab[19] and those found in a web search by using the term “recommended data repositories.” These lists have been compiled by the American Geophysical Union, BBSRC[20], BioSharing[21], COPDESS[22], DataMed[23], Elsevier[24], EMBO Press[25], F1000Research[26], GigaScience[27], NIH[28], PLOS[29], Scientific Data[30], Springer Nature/BioMed Central (both share the same list)[31], Web of Science[32], Wellcome Trust[33], and Wiley. All lists, including links to the online lists, were compiled into one list to compare recommendations (http://dx.doi.org/10.17632/zx2kcyvvwm.1). Not all data repositories indexed by the Web of Science’s Data Citation Index (DCI) were included as there is no publicly available list with all repositories indexed by the DCI, so retrieval of recommended repositories was done through an individual search. The repositories indexed by Re3Data were not included in our list of recommended repositories as Re3data functions as “a global registry of research data repositories”[34] and thus does not recommend repositories. However, Re3Data was used to verify the repository’s status, persistent identifiers, and obtained certifications.
Certified repositories
For our analysis of data repository certification schemes, we examined five certification schemes. These were the DSA, ICSU-WDS, NESTOR, TRAC, and ISO 16363/TDR. These schemes were chosen due to being used for certification (DSA, ICSU-WDS, NESTOR and ISO 16363/TDR) or as a self-assessment check for repositories (TRAC). The DSA[35], ICSU-WDS[36], and NESTOR[37] provide lists of certified repositories on their respective websites. ISO 16363/TDR certification has not yet been awarded.[38] We consulted the websites of the DSA, ICSU-WDS, NESTOR, TRAC, and ISO 16363/TDR certification schemes to see which repositories they certified. The results were compiled into one list (https://doi.org/10.17632/zx2kcyvvwm.1).
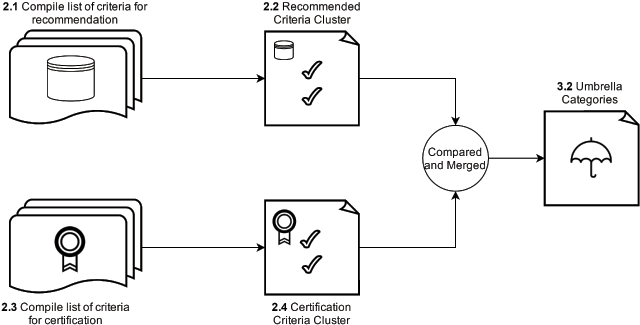
After composing the list of recommended repositories, we investigated which criteria are being used to determine a recommendation or certification, and whether an overlap exists between recommended and certified repositories, and the criteria used (Figure 1).
- 2.1 Compiled list of criteria for recommendation
- 2.2 Clustered criteria into the “Recommended Criteria Cluster”
- 2.3 Compiled list of criteria for certification
- 2.4 Clustered criteria into the “Certification Criteria Cluster”
- 3.2 Compared and merged steps 2.2 and 2.4 to create the umbrella categories
|
These steps will be discussed in turn.
Criteria used for recommendation and certification
Criteria for recommendation
To understand the motivation behind specific recommendations, we looked at the organizations’ selection criteria for their lists of recommended repositories. Four of the 17 organizations supplied criteria for this online alongside their lists: BioCADDIE, F1000Research, Scientific Data/Springer Nature (SD/SN), and Web of Science (WoS). The Research Data Alliance’s (RDA)[39] criteria for recommended repositories were also included in this analysis. Although the RDA does not maintain a list of recommended repositories, we included their criteria to balance out the weight between the number of organizations that recommend repositories and the number of organizations that provide criteria for certification. The criteria of the five organizations were then compiled into one list (https://doi.org/10.17632/zx2kcyvvwm.1).
Recommended Criteria Cluster
We categorized the criteria into 15 subheadings: Recognition, Mission, Transparency, Certification, Interface, Legal, Access, Structure, Retrievability, Preservation/Persistence, Curation, Persistent Identifier, Citability, Language, and Diversity of Data. These subheadings were derived from recurring and shared subjects throughout the different criteria lists. We then filtered out repetitions or criteria unique to one organization, namely Language and Diversity of Data, to create the “Recommended Criteria Cluster.”
Criteria for certification
We consulted relevant websites to obtain the criteria used by the DSA, ICSU-WDS, NESTOR, TRAC and ISO 16363/TDR certification schemes and compiled a list of all certification criteria of the five schemes. In the case of the DSA[40], ICSU-WDS[17], and NESTOR[41], these were found through their respective websites. The criteria for TRAC were found through the website of the Center for Research Libraries (CRL)[42], and the criteria for the ISO 16363/TDR were found through the Primary Trustworthy Digital Repository Authorisation Body, on the website of the Consultative Committee for Space Systems.[18]
References
- ↑ Piwowar, H.A.; Vision, T.J. (2013). "Data reuse and the open data citation advantage". PeerJ 1: e175. doi:10.7717/peerj.175. PMC PMC3792178. PMID 24109559. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3792178.
- ↑ Piwowar, H.A.; Day, R.S.; Fridsma, D.B. (2007). "Sharing detailed research data is associated with increased citation rate". PLoS One 2 (3): e308. doi:10.1371/journal.pone.0000308. PMC PMC1817752. PMID 17375194. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1817752.
- ↑ "NIH Grants Policy Statement" (PDF). National Institutes of Health. 2015. https://grants.nih.gov/grants/policy/nihgps/nihgps.pdf. Retrieved 27 January 2017.
- ↑ "Policy on data, software and materials management and sharing". Wellcome Trust. https://wellcome.ac.uk/funding/managing-grant/policy-data-software-materials-management-and-sharing. Retrieved 27 January 2017.
- ↑ Borgman, C.L. (2012). "The conundrum of sharing research data". Journal of the American Society for Information Science and Technology 63 (6): 1059-1078. doi:10.1002/asi.22634.
- ↑ Mayernik, M.S.; Callaghan, S.; Leigh, R. et al. (2014). "Peer Review of Datasets: When, Why, and How". Bulletin of the American Meteorological Society 96 (2): 191–201. doi:10.1175/BAMS-D-13-00083.1.
- ↑ 7.0 7.1 Dobratz, S.; Rödig, P.; Borghoff, U.M. et al. (2010). "The Use of Quality Management Standards in Trustworthy Digital Archives". International Journal of Digital Curation 5 (1): 46–63. doi:10.2218/ijdc.v5i1.143.
- ↑ Assante, M.; Candela, L.; Castelli, D. et al. (2016). "Are Scientific Data Repositories Coping with Research Data Publishing?". Data Science Journal 15 (6): 1–24. doi:10.5334/dsj-2016-006.
- ↑ Merson, L.; Gaye, O.; Guerin, P.J. (2016). "Avoiding Data Dumpsters--Toward Equitable and Useful Data Sharing". New England Journal of Medicine 374 (25): 2414-5. doi:10.1056/NEJMp1605148. PMID 27168351.
- ↑ McQuilton, P.; Gonzalez-Beltran, A.; Rocca-Serra, P. et al. (2016). "BioSharing: Curated and crowd-sourced metadata standards, databases and data policies in the life sciences". Database 2016: baw075. doi:10.1093/database/baw075. PMC PMC4869797. PMID 27189610. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4869797.
- ↑ Wilkinson, M.D.; Dumontier, M.; Aalbersberg, I.J. et al. (2016). "The FAIR Guiding Principles for scientific data management and stewardship". Scientific Data 3: 160018. doi:10.1038/sdata.2016.18. PMC PMC4792175. PMID 26978244. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4792175.
- ↑ "The 'FAIR' principles for scientific data management". UK Data Service. 8 June 2016. https://www.ukdataservice.ac.uk/news-and-events/newsitem/?id=4615. Retrieved 28 October 2016.
- ↑ "Guidelines on FAIR Data Management in Horizon 2020" (PDF). European Commission. 26 July 2016. http://ec.europa.eu/research/participants/data/ref/h2020/grants_manual/hi/oa_pilot/h2020-hi-oa-data-mgt_en.pdf. Retrieved 27 October 2016.
- ↑ "Big Data to Knowledge". National Institutes of Health. 2017. https://commonfund.nih.gov/bd2k.
- ↑ "Data Seal of Approval: About". DSA Board. https://www.datasealofapproval.org/en/information/about/. Retrieved 18 January 2017.
- ↑ 16.0 16.1 Dillo, I.; de Leeuw, L. (2015). "Ten Years Back, Five Years Forward: The Data Seal of Approval". International Journal of Digital Curation 10 (1): 363. doi:10.2218/ijdc.v10i1.363.
- ↑ 17.0 17.1 "Certification of WDS Members" (PDF). ICSU World Data System. 11 June 2012. https://www.icsu-wds.org/files/wds-certification-summary-11-june-2012.pdf. Retrieved 28 October 2016.
- ↑ 18.0 18.1 "Audit and Certification of Trustworthy Digital Repositories" (PDF). CCSDS. September 2011. https://public.ccsds.org/pubs/652x0m1.pdf. Retrieved 28 October 2016.
- ↑ "Recommendations". FAIRsharing.org. University of Oxford. https://fairsharing.org/recommendations/.
- ↑ "Resources". BBSRC. http://www.bbsrc.ac.uk/research/resources/. Retrieved 27 January 2017.
- ↑ "Databases". FAIRsharing.org. University of Oxford. https://fairsharing.org/databases/?q=&selected_facets=recommended:true.
- ↑ "Search for Repositories". COPDESS. https://copdessdirectory.osf.io/search/. Retrieved 27 January 2017.
- ↑ "Repository List". bioCADDIE. https://datamed.org/repository_list.php. Retrieved 27 January 2017.
- ↑ "Supported Data Repositories". Elsevier. https://www.elsevier.com/authors/author-services/research-data/data-base-linking/supported-data-repositories. Retrieved 27 January 2017.
- ↑ "Data Deposition". Author Guidelines. EMBO Press. http://msb.embopress.org/authorguide#datadeposition. Retrieved 27 January 2017.
- ↑ "Data Guidelines". How to Publish. F1000 Research. https://f1000research.com/for-authors/data-guidelines. Retrieved 27 January 2017.
- ↑ "Editorial Policies & Reporting Standards". Oxford University Press. https://academic.oup.com/gigascience/pages/editorial_policies_and_reporting_standards. Retrieved 27 January 2017.
- ↑ "NIH Data Sharing Repositories". U.S. National Library of Medicine. https://www.nlm.nih.gov/NIHbmic/nih_data_sharing_repositories.html. Retrieved 27 January 2017.
- ↑ "Data Availability". PLOS. http://journals.plos.org/plosbiology/s/data-availability. Retrieved 27 January 2017.
- ↑ "Recommended Data Repositories". Scientific Data. Macmillan Publishers Limited. https://www.nature.com/sdata/policies/repositories. Retrieved 27 January 2017.
- ↑ "Recommended Repositories". Springer Nature. Springer-Verlag GmbH. http://www.springernature.com/gp/authors/research-data-policy/repositories/12327124?countryChanged=true. Retrieved 27 January 2017.
- ↑ "Master Data Repository List". Web of Science. Clarivate Analytics. http://wokinfo.com/cgi-bin/dci/search.cgi. Retrieved 27 January 2017.
- ↑ "Data repositories and database resources". Wellcome Trust. https://wellcome.ac.uk/funding/managing-grant/data-repositories-and-database-resources. Retrieved 27 January 2017.
- ↑ "About". re3data.org. Karlsruhe Institute of Technology. https://www.re3data.org/about. Retrieved 27 January 2017.
- ↑ "Assessment". Data Seal of Approval. DSA Board. https://www.datasealofapproval.org/en/assessment/. Retrieved 20 February 2017.
- ↑ "Membership". ICSU World Data System. https://www.icsu-wds.org/community/membership/regular-members. Retrieved 20 February 2017.
- ↑ "Nestor Seal for Trustworthy Digital Archives". nestor-Geschäftsstelle. http://www.langzeitarchivierung.de/Subsites/nestor/EN/Siegel/siegel_node.html. Retrieved 20 February 2017.
- ↑ Larrimer, N. (20 January 2016). "Accreditation Program for ISO 16363 Trustworthy Digital Repositories Management Systems" (PDF). ANAB Heads Up. https://anab.qualtraxcloud.com/ShowDocument.aspx?ID=4446. Retrieved 28 October 2016.
- ↑ "Repository Bundle". Research Data Alliance. https://www.rd-alliance.org/group/data-fabric-ig/wiki/repository-bundle.html. Retrieved 27 January 2017.
- ↑ "Requirements". Data Seal of Approval. DSA Board. https://www.datasealofapproval.org/en/information/requirements/. Retrieved 07 April 2017.
- ↑ "Explanatory notes on the nestor Seal for Trustworthy Digital Archives". nestor Certification Working Group. July 2013. https://d-nb.info/1047613859/34. Retrieved 07 April 2017.
- ↑ OCLC and CRL (February 2007). "Trustworthy Repositories Audit & Certification: Criteria and Checklist" (PDF). Center for Research Libraries. http://www.crl.edu/sites/default/files/d6/attachments/pages/trac_0.pdf. Retrieved 07 April 2017.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. In some cases important information was missing from the references, and that information was added. The original article lists references alphabetically, but this version — by design — lists them in order of appearance. The BioSharing website has since become the "FAIRSharing" website, and as such the original BioSharing links point to the new website. Several other website URLs have also changed, and the updated URL is used here. The original includes several inline citations that are not listed in the references section; they have been omitted here.