Difference between revisions of "LII:Elements of Laboratory Technology Management"
Shawndouglas (talk | contribs) (Saving and adding more.) |
Shawndouglas (talk | contribs) (Saving and adding more.) |
||
| Line 248: | Line 248: | ||
* convert laboratory methods to fully automated systems; | * convert laboratory methods to fully automated systems; | ||
* deliberately design equipment and control, acquisition, and processing systems to meet the needs of this kind of application; | * deliberately design and manage equipment and control, acquisition, and processing systems to meet the needs of this kind of application; | ||
* train people to work in a more complex environment than they had been; and | * train people to work in a more complex environment than they had been; and | ||
* build the automation infrastructure (i.e., interfacing and data standards) needed to make these systems realizable and effective, without taking on significant cost. | * build the automation infrastructure (i.e., interfacing and data standards) needed to make these systems realizable and effective, without taking on significant cost. | ||
| Line 258: | Line 258: | ||
This doesn’t diminish the science used in laboratory work; rather, it adds a level of sophistication that hasn’t been widely dealt with: thinking beyond the basic science process to its implementation in a continuous automated system. This is a much more complex undertaking since 1. data will be created at a high rate and 2. we want to be sure that this is high-quality data and not just the production of junk at a high rate. | This doesn’t diminish the science used in laboratory work; rather, it adds a level of sophistication that hasn’t been widely dealt with: thinking beyond the basic science process to its implementation in a continuous automated system. This is a much more complex undertaking since 1. data will be created at a high rate and 2. we want to be sure that this is high-quality data and not just the production of junk at a high rate. | ||
This type of thinking is not limited to quality control work. It can be readily applied to research as well, where economical high-volume experiments can be used to support statistical experimental design methodologies and more exhaustive sample processing, as well as today’s screening applications in life sciences. It is also readily applied to environmental monitoring and regulatory evaluations. And while this kind of thinking may be new to scientific applications, it isn’t new technology. Work that has been done in automated manufacturing can serve as a template for the work that has to be done in laboratory process automation. | |||
Let's return to the first two bullets in this section: laboratory method conversion and system management and planning. If you gave four labs a method description and asked them to automate it, you’d get varied implementations of four groups doing the same thing independently. If we are going to turn lab automation into the useful tool it can be, we need to take a different approach: cooperative development of automated systems. | |||
In order to be useful, a description of a fully automated system needs more that a method description. It needs equipment lists, source code, etc. in sufficient detail that you can purchase the equipment needed and put the system together expecting it work. However, we can do better than that. In a given industry, where labs are doing the same testing on the same types of samples, we should be able to have them come together and designate and test automated systems to meet the need. Once that is done, vendors can pick up the description and be able to build products suitable to carry out the analysis or test. The problem labs face is getting the work done at a reasonable cost. If there isn’t a competitive advantage to having a unique test, cooperate so that standardized modules for testing can be developed. | |||
This changes the process of lab automation from a build-it-from-scratch mentality to the planned connection of standardized automated components into a functioning system. | |||
===Experimental methods=== | |||
There is relatively little that can be said about experimental methods at this point. Aside from the clinical industry, not enough work has been done to give really good examples of intentionally designed automated systems that can be purchased, installed in a lab, and expected to function.{{Efn|Having a data system connected to, or in control of, a process is not the same as full automation. For example, there are automated Karl Fisher systems (for water analysis), but they only address titration activities and not sample preparation. A vendor can only take things so far in commercial products unless labs describe a larger role for automation, one that will vary by application. The point is we need a formalized description of that larger context.}} There are some examples, including ELIZA robotics analysis systems from Caliper Life Sciences and Pressurized Liquid Extraction Systems from Fluid Management Systems. Many laboratory methods are designed with the assumption that people will be doing the work (manually), and any addition of automation would require conversion of that method (Figure 9). | |||
[[File:Fig9 Liscouski ElementsLabTechMan14.png|800px]] | |||
{{clear}} | |||
{| | |||
| STYLE="vertical-align:top;"| | |||
{| border="0" cellpadding="5" cellspacing="0" width="800px" | |||
|- | |||
| style="background-color:white; padding-left:10px; padding-right:10px;"| <blockquote>'''Figure 9.''' The consideration of a lab's experimental methods for laboratory technology management</blockquote> | |||
|- | |||
|} | |||
|} | |||
What we need to begin doing is looking at the development of automated methods as a distinct task similar to the published manual methods by [[ASTM International]] (ASTM), the [[Environmental Protection Agency]] (EPA), and the United States Pharmacopeia (USP), with the difference being that automation is not viewed as mimicking human actions but as well-designed and optimized systems that support the science and any associated production processes (i.e., a scientific manufacturing implementation that includes integration with informatics systems). We need to think “bigger,” without limiting our vision to just the immediate task but rather looking at how it fits into lab-wide operations. | |||
===Lab-specific technologies and information technology=== | |||
This section quickly covers the next two elements of laboratory technology management at the same time, as many aspects of these elements have already been covered elsewhere. See Figure 10 and Figure 11 for a deeper dive into these two elements.{{Efn|For more information about lab-specific technologies, please refer to ''[https://www.researchgate.net/publication/275351757_Computerized_Systems_in_the_Modern_Laboratory_A_Practical_Guide Computerized Systems in the Modern Laboratory: A Practical Guide]''.}} | |||
[[File:Fig10 Liscouski ElementsLabTechMan14.png|1000px]] | |||
{{clear}} | |||
{| | |||
| STYLE="vertical-align:top;"| | |||
{| border="0" cellpadding="5" cellspacing="0" width="1000px" | |||
|- | |||
| style="background-color:white; padding-left:10px; padding-right:10px;"| <blockquote>'''Figure 10.''' Reviewing the many aspects of lab-specific technologies for laboratory technology management</blockquote> | |||
|- | |||
|} | |||
|} | |||
[[File:Fig11 Liscouski ElementsLabTechMan14.png|800px]] | |||
{{clear}} | |||
{| | |||
| STYLE="vertical-align:top;"| | |||
{| border="0" cellpadding="5" cellspacing="0" width="800px" | |||
|- | |||
| style="background-color:white; padding-left:10px; padding-right:10px;"| <blockquote>'''Figure 11.''' Consideration of information technologies for laboratory technology management</blockquote> | |||
|- | |||
|} | |||
|} | |||
There is one point that needs to be made with respect to information technologies. While lab managers do not need to understand the implementation details of those technologies, they do need to be aware of the potential they offer in the development of a structure for laboratory automation implementations. Management is responsible for lab automation planning, including choosing the best technologies; in other words, management must manage the “big picture” of how technologies are used to meet their lab's purpose. | |||
In particular, managers should pay close attention to the role of client-server systems and virtualization, since they offer design alternatives that impact the choice of products and the options for managing technology. This is one area where good relationships with IT departments are essential. We’ll be addressing these and other information technologies in more detail in other publications. | |||
===Systems integration=== | |||
Revision as of 19:21, 16 February 2021
Title: Elements of Laboratory Technology Management
Author for citation: Joe Liscouski, with editorial modifications by Shawn Douglas
License for content: Creative Commons Attribution 4.0 International
Publication date: 2014
|
|
This article should be considered a work in progress and incomplete. Consider this article incomplete until this notice is removed. |
Introduction
This discussion is less about specific technologies than it is about the ability to use advanced laboratory technologies effectively. “Effectively” means that products and technologies are used successfully to address needs in your lab, and that they improve the lab’s ability to function. If they don't do that, you’ve wasted your money. Additionally, if the technology in question hasn’t been deployed according to a deliberate plan, your funded projects may not achieve everything they could. Optimally, when applied thoughtfully, the available technologies should result in the transformation of lab work from a labor-intensive effort to one that is intellectually intensive, making the most effective use of people and resources.
People come to the subject of laboratory automation from widely differing perspectives. To some it’s about robotics, to others it’s about laboratory informatics, and even others view it as simply data acquisition and analysis. It all depends on what your interests are, and more importantly what your immediate needs are.
People began working in this field in the 1940s and 1950s, with the work focused on analog electronics to improve instrumentation; this was the first phase of lab automation. Most notably were the development of scanning spectrophotometers and process chromatographs. Those who first encountered this equipment didn’t think much of it and considered it the world as it’s always been. Others who had to deal with products like the Spectronic 20[a] (a single-beam manual spectrophotometer), and use it to develop visible spectra one wavelength measurement at a time, appreciated the automation of scanning instruments.
Mercury switches and timers triggered by cams on a rotating shaft provided chromatographs with the ability to automatically take samples, actuate back flush valves, and take care of other functions without operator intervention. This left the analyst with the task of measuring peaks, developing calibration curves, and performing calculations, at least until data systems became available.
The direction of laboratory automation changed significantly when computer chips became available. In the 1960s, companies such as PerkinElmer were experimenting with the use of computer systems for data acquisition as precursors to commercial products. The availability of general-purpose computers such as the PDP-8 and PDP-12 series (along with the Lab 8e) from Digital Equipment, with other models available from other vendors, made it possible for researchers to connect their instruments to computers and carry out experiments. The development of microprocessors from Intel (4004, 8008) led to the evolution of “intelligent” laboratory equipment ranging from processor-controlled stirring hot-plates to chromatographic integrators.
As researchers learned to use these systems, their application rapidly progressed from data acquisition to interactive control of the experiments, including data storage, analysis, and reporting. Today, the product set available for laboratory applications includes data acquisition systems, laboratory information management systems (LIMS), electronic laboratory notebooks (ELNs), laboratory robotics, and specialized components to help researchers, scientists, and technicians apply modern technologies to their work.
While there is a lot of technology available, the question remains "how do you go about using it?" Not only do we need to know how to use it, but we also must do so while avoiding our own biases about how computer systems operate. Our familiarity with using computer systems in our daily lives may cause us to assume they are doing what we need them to do, without questioning how it actually gets done. “The vendor knows what they are doing” is a poor reason for not testing and evaluating control parameters to ensure they are suitable and appropriate for your work.
Moving from lab functions and requirements to practical solutions
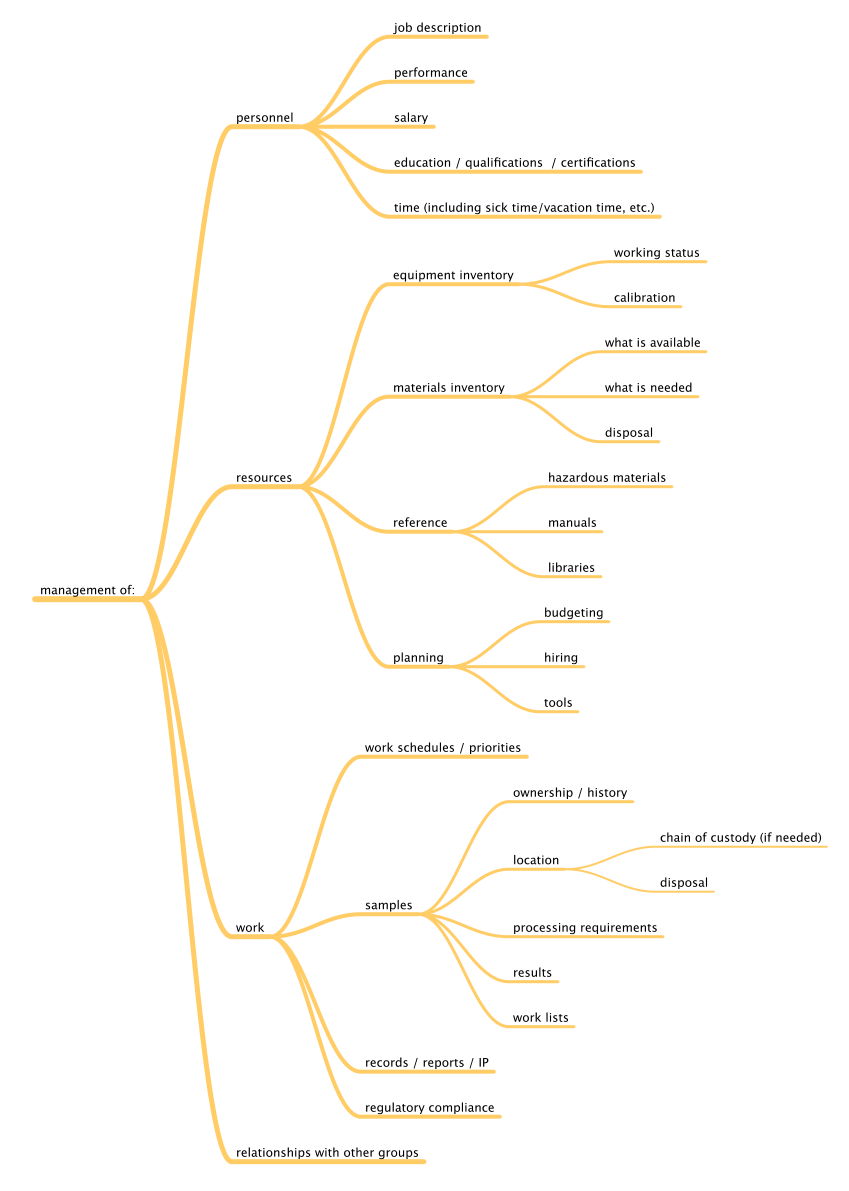
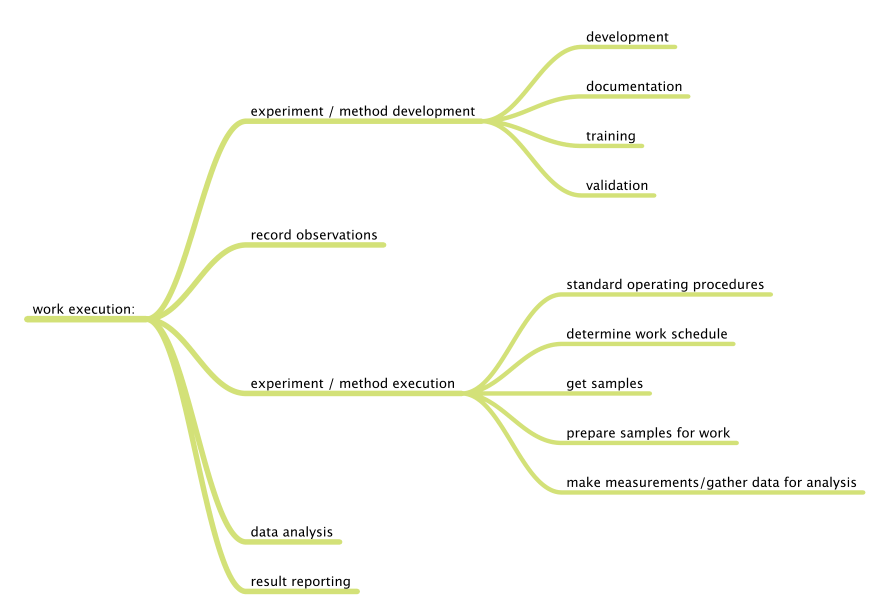
Before we can begin to understand the application of the tools and technologies that are available, we have to know what we want to accomplish, specifically what problems we want to solve. We can divide laboratory functions into two broad classes: management and work execution. Figure 1 addresses management functions, wheras Figure 2 addresses work execution functions, all common to laboratories. You can add to them based on your own experience.
|
|
Vendors have been developing products to address these work areas, and there are a lot of products available. Many of them are "point" solutions: products that are focused on one aspect of work without an effort to integrate them with others. That isn’t surprising since there isn’t an architectural basis for integration aside from specific hardware systems (e.g., Firewire, USB) or vendor-specific software systems (e.g., office product suites). Another issue in scientific work is that the vendor may only be interested in solving a particular problem, with most of the emphasis on an instrument or technique. They may provide the software needed to support their hardware, with data transfer and integration left to the user.
As you work through this document, you’ll find a map of management responsibilities and technologies. How do you connect the above map of functions to the technologies? Applying software and hardware solutions to your lab's needs requires deliberate planning. The days of purchasing point solutions to problems have passed. Today's lab managers need to think more broadly about product usage and how components of lab software systems work together. The point of this document is to help you understand what you need to think about in that regard.
Given those summaries of lab activities, how do we apply available technologies to improve lab operations? Most of the answers fall under the heading of "laboratory automation," so we’ll begin by looking at what that is.
What is laboratory automation?
This isn’t a trivial question; your answer may depend on the field you are working in, your experience, and your current interests. To some it means robotics, to others it is a LIMS (or their clinical counterpart, the laboratory information systems or LIS). The ELN and instrument data systems (IDS) are additional elements worth noting. These are examples of product classes and technologies used in lab automation, but they don’t define the field. Wikipedia provides the following as a definition[1]:
Laboratory automation is a multi-disciplinary strategy to research, develop, optimize and capitalize on technologies in the laboratory that enable new and improved processes. Laboratory automation professionals are academic, commercial and government researchers, scientists and engineers who conduct research and develop new technologies to increase productivity, elevate experimental data quality, reduce lab process cycle times, or enable experimentation that otherwise would be impossible. The most widely known application of laboratory automation technology is laboratory robotics. More generally, the field of laboratory automation comprises many different automated laboratory instruments, devices, software algorithms, and methodologies used to enable, expedite and increase the efficiency and effectiveness of scientific research in laboratories.
And McDowall offered this definition in 1993[2]:
Apparatus, instrumentation, communications or computer applications designed to mechanize or automate the whole or specific portions of the analytical process in order for a laboratory to provide timely and quality information to an organization
These definitions emphasize equipment and products, and that is where typical approaches to lab automation and the work we are doing part company. Products and technologies are important, but what is more important is figuring out how to use them effectively. The lack of consistent success in the application of lab automation technologies appears to stem from this focus on technologies and equipment—“what will this product do for my lab?”—rather than methodologies for determining what is needed and how to implement solutions.
Having a useful definition of laboratory automation is crucial since how we approach the work depends on how we see the field developing. The definition the Institute for Laboratory Automation (ILA) bases its work on is this:
Laboratory automation is the process of determining needs and requirements, planning projects and programs, evaluating products and technologies, and developing and implementing projects according to a set of methodologies that results in successful systems that increase productivity, improve the effectiveness and efficiency of laboratory operations, reduce operating costs, and provide higher-quality data.
The field includes the use of data acquisition, analysis, robotics, sample preparation, laboratory informatics, information technology, computer science, and a wide range of technologies and products from widely varying disciplines, used in the implementation of projects.
Why "process" is important
Lab automation isn’t about stuff, but how we use stuff. The “process” component of the ILA definition is central to what we do. To quote Frank Zenie[b] (one of the founders of Zymark Corporation) “you don’t automate a thing, you automate a process.” You don’t automate an instrument, you automate the process of using one. Autosamplers are a good example of successful automation: they address the process of selecting a sample vial, withdrawing fluid, positioning a needle into an injection port, injecting the sample, preparing the syringe for the next injection, and indexing the sample vials when needed.
A number of people have studied the structure of science and the relationship between disciplines. Lab automation is less about science and more about how the work of science is done. Before lab automation can be considered for a project, the underlying science has to be done. In other words, the process that automation is going to be applied to has to exist first. It also has to be the right process for consideration. This is a point that needs attention.
If you are going to spend resources on a project, you have to make sure you have a well-characterized process and that the process is both optimal and suitable for automation. This means:
- The process is well documented, people that use the process have been trained on that documented process, and their work is monitored to determine any shortcuts, workarounds, or other variations from that documented process. Differences between the published process and the one actually in use can have a significant impact on the success of a particular project design.
- The process’s “readiness for automation” has been determined. The equipment used is suitable for automation or the changes needed to make it suitable are known, and it can be done at reasonable cost. Any impact on warranties has been determined and found to be acceptable.
- If several process options exist (e.g., different test protocols for the same test question), they are evaluated for their ability to meet the needs of the science and to be successfully implemented. Other options, such as outsourcing, should be considered to make the best use of resources; is may be more cost-effective to outsource than automate.
When looking at laboratory processes, it's useful to recognize they may operate on different levels. There may be high-level processes that address the lab's reason for existence and cover the mechanics of how the lab functions, as well as low-level processes that address individual functions of the lab. This process view is important when we consider products and technologies, as the products have to fit the process, the general basis for product requirements. Discussions often revolve around LIMS and ELNs are one example, with questions being asked about whether one or both are required in the lab, or whether an ELN can replace a LIMS.
These and other similar questions reflect both vendor influence and a lack of understanding of the technologies and their application. Some differentiate the two types of technology based on “structured” (as found in LIMS) vs. “unstructured” (as found in ELNs) data. Broadly speaking, LIMS come with a well-defined, extensible, database structure, while ELNs are viewed as unstructured since you can put almost anything into an ELN and organize the contents as you see fit. But this characterization doesn’t work well either since as a user I might consider the contents, along with an index, as having a structure. This is more of an information technology approach rather than one that addresses laboratory needs. In the end, an understanding of lab processes is still required to resolve most issues.
LIMS are well-defined entities[c] and the only one of the two to carry an objective industry-standard description. LIMS are also designed to manage the processes surrounding laboratory testing in a wide variety of industries (e.g., from analytical, physical, and environmental testing, to clinical testing, which is usually associated with the LIS). The lab behavior process model is essentially the same across industries and disciplines. Basically, if you are running an analytical lab and need to manage samples and test results while answering questions about the status of testing on a larger scale than what you can memorize[d], a LIMS is a good answer.
At the time of this writing, there is no standardized definition of an ELN, though a forthcoming update of ASTM E1578 intends to recitfy that. However, given the current hype about ELNs, any product with that designation is going to get noticed. Lets avoid the term and replace it with a set of software types that addresses similar functionality:
1. Scripted execution systems – These are software systems that guide an analyst in the conduct of a procedure (process); examples include Velquest (now owned by Accelrys) products and the scripting notebook function (vendor's description) in [LabWare, Inc.|LabWare LIMS]].
2. Journal or diary system - These are software systems that provide a record of laboratory work; a word processing system might fill this need, although there are products with features specifically designed to assist lab work.
3. Application- or discipline-specific record keeping systems – These are software systems designed for biology, chemistry, mathematics and other areas that contain features that allow you to record data and text in a variety of forms that are geared toward the needs of specific areas of science.
This is not an exhaustive list of forms or functionality, but it is sufficient to make a point. The first, scripted execution, is designed around a process or, more specifically, designed to give the user a mechanism to describe the sequential steps in a process so that they can be repeated under strict controls. These do not replace a LIMS but can be used synergistically with one, or with software that duplicates LIMS capabilities (some have suggested enterprise resource planning or ERP systems as a substitute). The other two types are repositories of lab information: equations, data, details of procedures, etc. There is no general underlying process as there is with LIMS. They can provide a researcher with a means of describing experiments, collecting data, and performing analyses, which you can correctly view as processes, but they are unique to that researcher or lab and not based on any generalized industry model, as we see in testing labs.
Why is this important? These descriptions illustrate something fundamental: process dictates needs, and needs set requirements for products and technology. The “do we need a LIMS or ELN” question is meaningless without an understanding of the processes that operate in your laboratory.
The role of processes in integration
From the standpoint of equipment, laboratories are often a collection of instruments, computer systems, sample preparation stations, and other test or measurement facilities. One goal frequently stated by lab managers is that “ideally we’d like all this to be integrated.” The purpose of integration is to streamline the operations, reduce human labor, and provide a more efficient way of doing work. You are integrating the equipment and systems used to execute one or more processes. However, without a thorough evaluation of the processes in the lab, there is no basis for integration.
Highlighting the connection between processes and integration, we see why defining what laboratory automation is remains important. One definition can lead to purchasing products and limiting the scope of automation to individual tasks. Another will take you through an evaluation of how your lab works, how you want it to work, and how to produce a framework for getting you there.
The elements of laboratory technology management
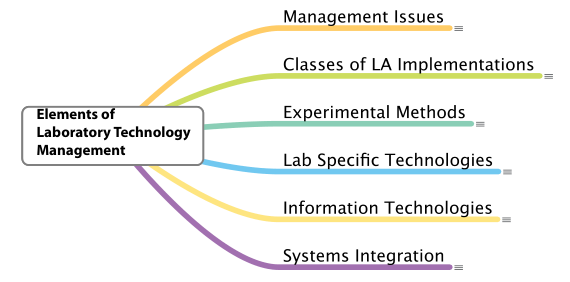
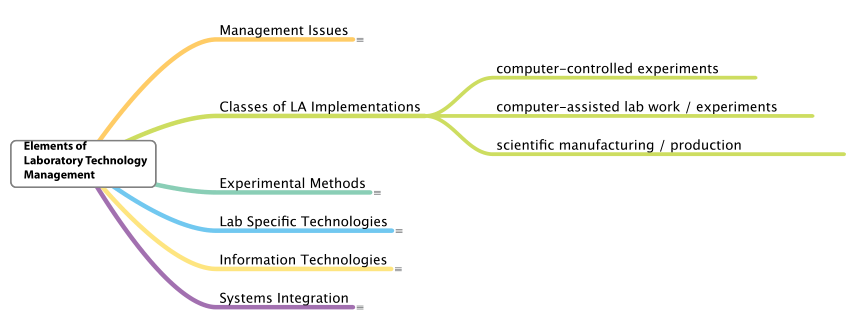
If lab automation is a process, we need to look at the elements that can be used to make that process work. The first thing that is needed is a structure that both shows how elements of lab automation relate to each other and act as guides for someone coming into the field. That structure also serves as a framework for organizing knowledge about the field. The major elements of the structure are shown in Figure 3.
|
Why is it important to recognize these laboratory automation management elements? As automation technology sees broader appeal across multiple industries, there is an inevitability to the use of automation technologies in labs. Vendors are putting chips and programmed intelligence into every product with the goal of making them easier to use, while reducing the role of human judgment (which can lead to an accumulation of errors in tasks like pippetting) and the amount of work people have to do to get work done. There are few negatives to this philosophy, aside from one point: if we don’t understand how these systems are working and haven’t planned for their use, we won’t get the most benefit from them, and we may accept results from systems blindly without really questioning their suitability and the results they produce. One of the main points of this work is that the use of automation technologies should be planned and managed. That brings us to the first major element: management issues.
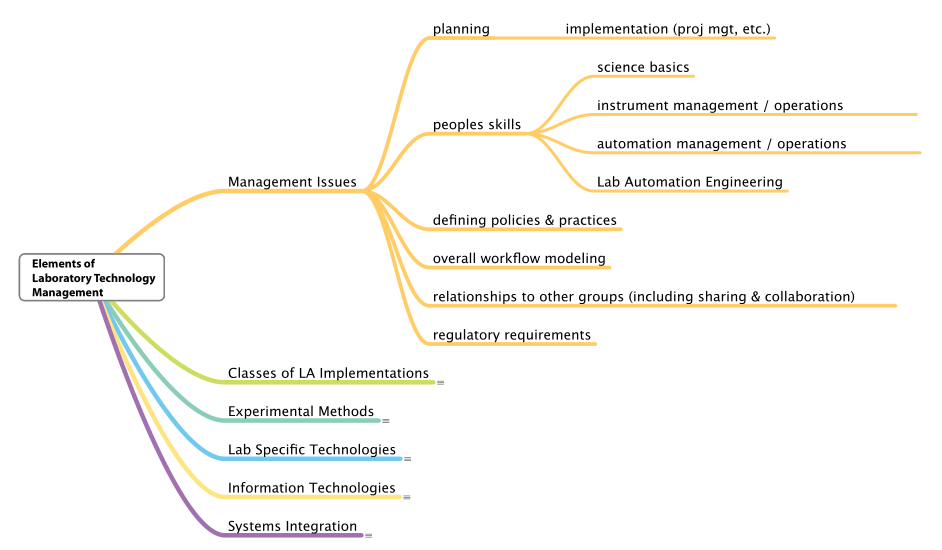
Management issues
The first thing we need to address is who “management” is. Unless the lab is represented solely by you, there are presumably layers of management. Depending on the size of the organization, this may mean one individual or a group of individuals who are responsible for various management aspects of the laboratory. Broadly speaking, those individuals will have a skillset that can appropriately address laboratory technology management, including project and program management, people skills, workflow modeling, and regulatory knowledge, to name a few (Figure 4). The need for these skillsets may vary slightly depending on the type of manager.
|
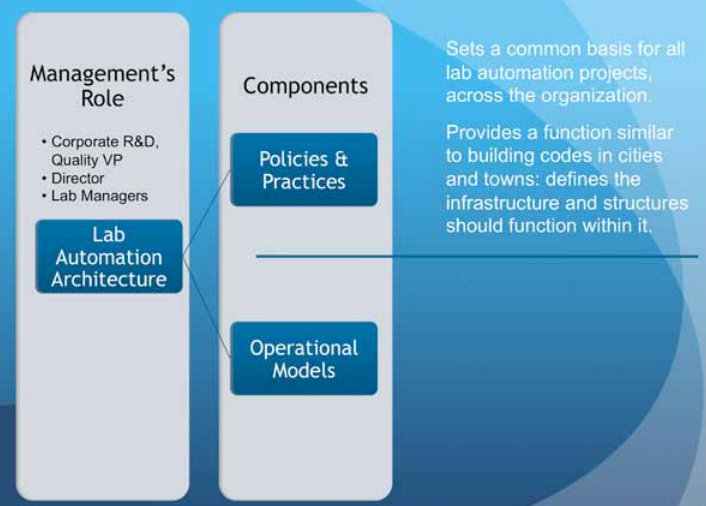
Aside from reviewing and approving programs ("programs" are efforts that cover multiple projects), “senior management” is responsible for setting the policies and practices that govern the conduct of laboratory programs. This is part of an overall architecture for managing lab operations, and has two components (Figure 5): setting policies and practices and developing operational models.[e]
|
Before the incorporation of informatics into labs, senior management’s involvement wasn’t necessary. However, the storage of intellectual property in electronic media has forced a significant change in lab work. Before informatics, labs used to be isolated from the rest of the organization, with formal contact made through the delivery of results, reports, and presentations. The desire for more effective, streamlined, and integrated information technology operations, and the development of information technology support groups, means that labs are now also part of the corporate picture. In organizations that have multiple labs, more efficient use of resources results in a desire to reduce duplication of work. You might easily justify two labs having their own spectrophotometers, but duplicate LIMS doing the same thing is going to require some explaining.
With the addition of informatics in the lab, management involvement is critical to support the success of laboratory programs. Among the reasons often cited for failure of laboratory programs is the lack of management involvement and, by extension, a lack of oversight, though these failures are usually stated without clarifying what management involvement should actually look like. To be clear, senior management's role is to ensure that programs are conducted in a way that:
- are common across all labs, such that all programs are conducted according to the same set of guidelines;
- are well-designed, supportable, and can be upgraded;
- are consistent with good project management practices;
- are conducted in a way that allows the results to be reused elsewhere in the company;
- are well-documented; and
- lead to successful results.
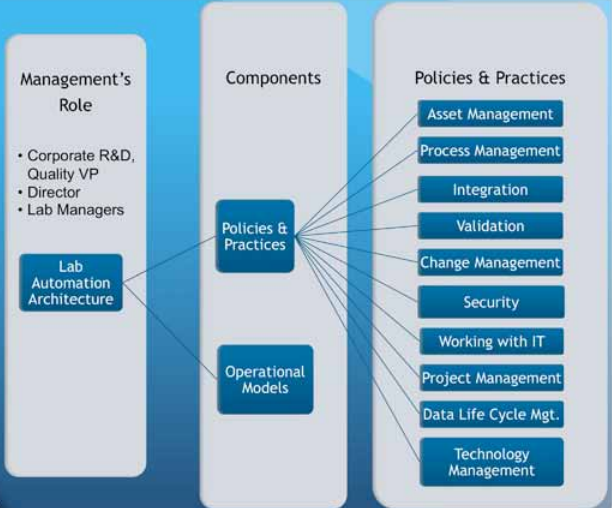
When work in lab automation began, it was usually the effort of one or two individuals in a lab or company. Today, we need a cooperative effort from management, lab staff, IT support, and, if available, LAEs. One of the reasons management must establish policies and practices (Figure 6) is to enable people to effectively work together, so they are working from the same set of ground rules and expectations and producing consistent and accurate results.
|
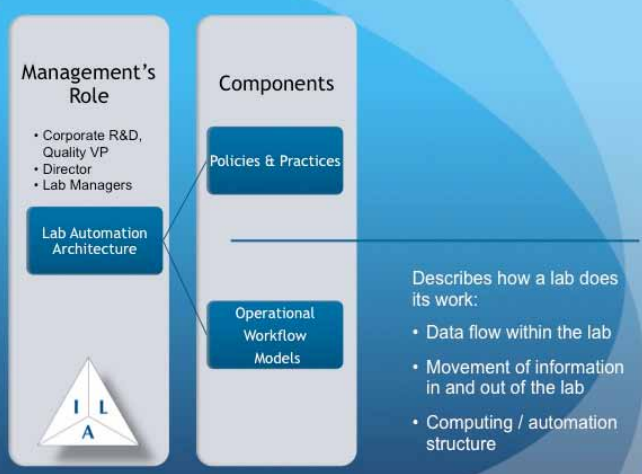
"Lab management" are responsible for understanding how their lab needs to operate in order to meet the lab's goals. Before automation became a factor, lab management’s primary concern was managing laboratory personnel and helping them get their work done. In the early stages of lab automation, the technologies were treated as add-ons that assisted personnel in getting work done. Today, lab managers need to move beyond that mindset and look at how their role must shift towards planning for and managing automation systems that take on more of the work in the lab. As a result, lab managers need to take on the role of technology planners in addition to managing people. The implementation of those plans may be carried out by others (e.g., laboratory automation engineers [LAEs], IT specialists), but defining the objectives and how the lab will function with a combination of people and systems is a task squarely in the lap of lab management, requiring the use of operational workflow models (Figure 7) to define the technologies and products suitable for their lab's work.
|
Problems can occur when different operational groups have conflicting sets of priorities. Take for example the case of labs and IT support. At one point, lab computing only affected the lab systems, but today's labs share resources with other groups, requiring "IT management" to ensure that work in one place doesn’t adversely impact another. Most issues around elements such as networks and security can be handled through implementing effective network design and bridges to isolate network segments when necessary. More often than not, problems are likely to occur in the choice and maintenance of products, and the policies IT implements to provide cost-effective support. Operating system upgrades are one place issues can occur if those changes cause products used in lab work to break because the vendor is slow in responding to OS changes. Another place that issues can occur is in product selection; IT managers may want to minimize the number of vendors it has to deal with and prefer products that use the same database system deployed elsewhere. That policy may adversely limit the products that the lab can choose from. From the lab's perspective, they need the best products in order to get their work done; from the IT group's perspective, they see it as driving up support costs. The way to avoid these issues, and others, is for senior managers to determine the priorities and keep the inter-department politics out of the discussion.
Classes of laboratory automation implementation
The second element of laboratory technology management to address is laboratory automation implementation. There are three classes of implementation to address (Figure 8):
- Computer-controlled experiments: This includes data collection in high-energy physics, LabView implemented systems, instrument command and control, and robotics. This implementation class involves systems where the computer is an integral part of the experiment, doing data collection and/or experiment control.
- Computer-assisted lab work/experiments: This includes work that could be done without a computer, but machines and software have been added to improve the process. Examples include chromatography data systems (CDS), ELNs used for documentation, and classic LIMS.
- Scientific manufacturing: This implementation class focuses on production systems, including high-throughput screening, lights-out lab automation, process analytical technologies, and quality by design (QbD) initiatives.
|
Computer-controlled experiments
This is where the second phase of laboratory automation began: when people began using digital computers with connections to their instrumentation. We moved in stages from simple data acquisition, acquiring a few points to show that we can accurately represent voltages from the equipment, to collecting multiple data streams over time and storing the results on disks. The next step consisted of automatically collecting the data, processing it, storing it, and reporting results from equipment such as chromatographs, spectrophotometers, and other devices.
Software development moved from assembly language programming to higher-level languages programming, and then specialized systems that provide a graphical interface to the programmer. Products like LabVIEW[f] allow the developer to use block diagrams to describe the programming and processing that have to be done, and provide the user with an attractive user interface with which to work. This is a far cry from embedding machine language programming in the BASIC language code, as was done in some earlier PC systems.
Robots offer another example of this class of work, where computers control the movement of equipment and materials through a process that prepares samples for analysis, and may include the analysis itself.
While commercial products have overtaken much of the work of interfacing, data acquisition, and data processing (in some cases, the instruments-computer combination are almost indistinguishable from the instruments themselves), the ability to deal with instrument interfacing and programming is still an essential skillset for those working in research and applications where commercial systems have yet to be developed.
It’s interesting that people often look at modern laboratory instrumentation and say that everything has gone digital. That’s far from the case. They may point to a single pan balance or thermometer with a digital readout as examples of a “digital” instrument, not realizing that the packaging contains sensors, analog-digital (A/D) converters, and computer control systems to manage the device and its communications. The appearance of a “digital” device masks what is going on inside; we still need to understand the transition from the analog world into the digital domain.
Computer-assisted lab work/experiments
There is a difference between work that can be done by computer and work that has to be done by computer; we just looked at the latter case. There’s a lot of work that goes on in laboratories that could be done and has been done by people, but in today’s labs we prefer to do that work with the aid of automated systems. For example, the management of samples in a testing laboratory used to be done by people logging samples in and keeping record books of the work that is scheduled and has been performed. Today, that work is better managed by a LIMS (or a LIS in the clinical world). The analysis of instrumental data used to be done by hand, and now it is more commonly done by instrument data systems that are faster, more accurate, and permit more complex analysis at a lower cost.
Do robots fit in this category? One could argue they are simply doing work that could be done manually performed in the past. The reason we might not consider robots in this class is that in many cases the equipment used for the robots is different than the equipment that’s used by human beings. As such, the two really aren’t interchangeable. If the LIMS or instrument data system were down, people could pick up the work manually; however, that may not be the case if a robot goes offline. It’s a small point and you can go either way on this.
The key element for this class of implementation is that the use of computers—and, if you prefer, robotics—is an option and not a requirement. Yet that option improves productivity, reduces cost, and provides better quality and more consistent data.
Scientific manufacturing
This isn’t so much a new implementation class as it is a formal recognition that much of what goes on in a laboratory mirrors work that’s done in manufacturing; some work in quality control is so routine that it matches assembly line work of the 1960s. The major difference, however, is that a lab's major production output is knowledge, information, and data (K/I/D). The work in this category is going to expand as a natural consequence of increasing automation, which must be addressed. If this is the direction things are going to go, then we need to do it right.
Recognizing this point has significant consequences. Rather than just letting things evolve, we can take advantage of the situation and drive situations that are appropriate for this level of automation into useful practice. This means we must:
- convert laboratory methods to fully automated systems;
- deliberately design and manage equipment and control, acquisition, and processing systems to meet the needs of this kind of application;
- train people to work in a more complex environment than they had been; and
- build the automation infrastructure (i.e., interfacing and data standards) needed to make these systems realizable and effective, without taking on significant cost.
In short, this means opening up another dimension to laboratory work as a natural evolution of work practices. If you look in the direction things are going in the lab, where large sample volume processing is necessary (e.g., high-throughput screening), it is simply a reflection of reality.
If we look at quality control applications and manufacturing processes, we basically see one production process (quality control or QC) layered on another (production), where ultimately the two merge into continuous production and testing. This is a logical conclusion to work described by process analytical technologies and quality-by-design.
This doesn’t diminish the science used in laboratory work; rather, it adds a level of sophistication that hasn’t been widely dealt with: thinking beyond the basic science process to its implementation in a continuous automated system. This is a much more complex undertaking since 1. data will be created at a high rate and 2. we want to be sure that this is high-quality data and not just the production of junk at a high rate.
This type of thinking is not limited to quality control work. It can be readily applied to research as well, where economical high-volume experiments can be used to support statistical experimental design methodologies and more exhaustive sample processing, as well as today’s screening applications in life sciences. It is also readily applied to environmental monitoring and regulatory evaluations. And while this kind of thinking may be new to scientific applications, it isn’t new technology. Work that has been done in automated manufacturing can serve as a template for the work that has to be done in laboratory process automation.
Let's return to the first two bullets in this section: laboratory method conversion and system management and planning. If you gave four labs a method description and asked them to automate it, you’d get varied implementations of four groups doing the same thing independently. If we are going to turn lab automation into the useful tool it can be, we need to take a different approach: cooperative development of automated systems.
In order to be useful, a description of a fully automated system needs more that a method description. It needs equipment lists, source code, etc. in sufficient detail that you can purchase the equipment needed and put the system together expecting it work. However, we can do better than that. In a given industry, where labs are doing the same testing on the same types of samples, we should be able to have them come together and designate and test automated systems to meet the need. Once that is done, vendors can pick up the description and be able to build products suitable to carry out the analysis or test. The problem labs face is getting the work done at a reasonable cost. If there isn’t a competitive advantage to having a unique test, cooperate so that standardized modules for testing can be developed.
This changes the process of lab automation from a build-it-from-scratch mentality to the planned connection of standardized automated components into a functioning system.
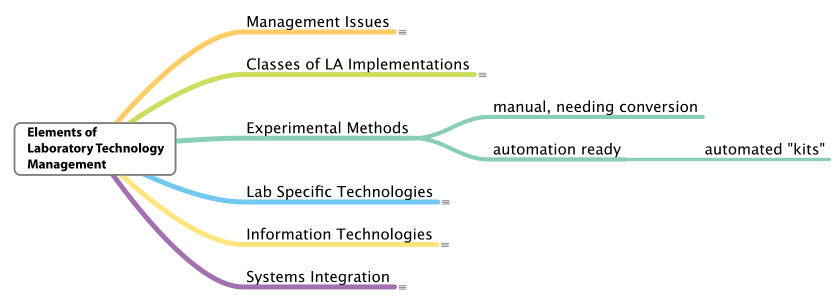
Experimental methods
There is relatively little that can be said about experimental methods at this point. Aside from the clinical industry, not enough work has been done to give really good examples of intentionally designed automated systems that can be purchased, installed in a lab, and expected to function.[g] There are some examples, including ELIZA robotics analysis systems from Caliper Life Sciences and Pressurized Liquid Extraction Systems from Fluid Management Systems. Many laboratory methods are designed with the assumption that people will be doing the work (manually), and any addition of automation would require conversion of that method (Figure 9).
|
What we need to begin doing is looking at the development of automated methods as a distinct task similar to the published manual methods by ASTM International (ASTM), the Environmental Protection Agency (EPA), and the United States Pharmacopeia (USP), with the difference being that automation is not viewed as mimicking human actions but as well-designed and optimized systems that support the science and any associated production processes (i.e., a scientific manufacturing implementation that includes integration with informatics systems). We need to think “bigger,” without limiting our vision to just the immediate task but rather looking at how it fits into lab-wide operations.
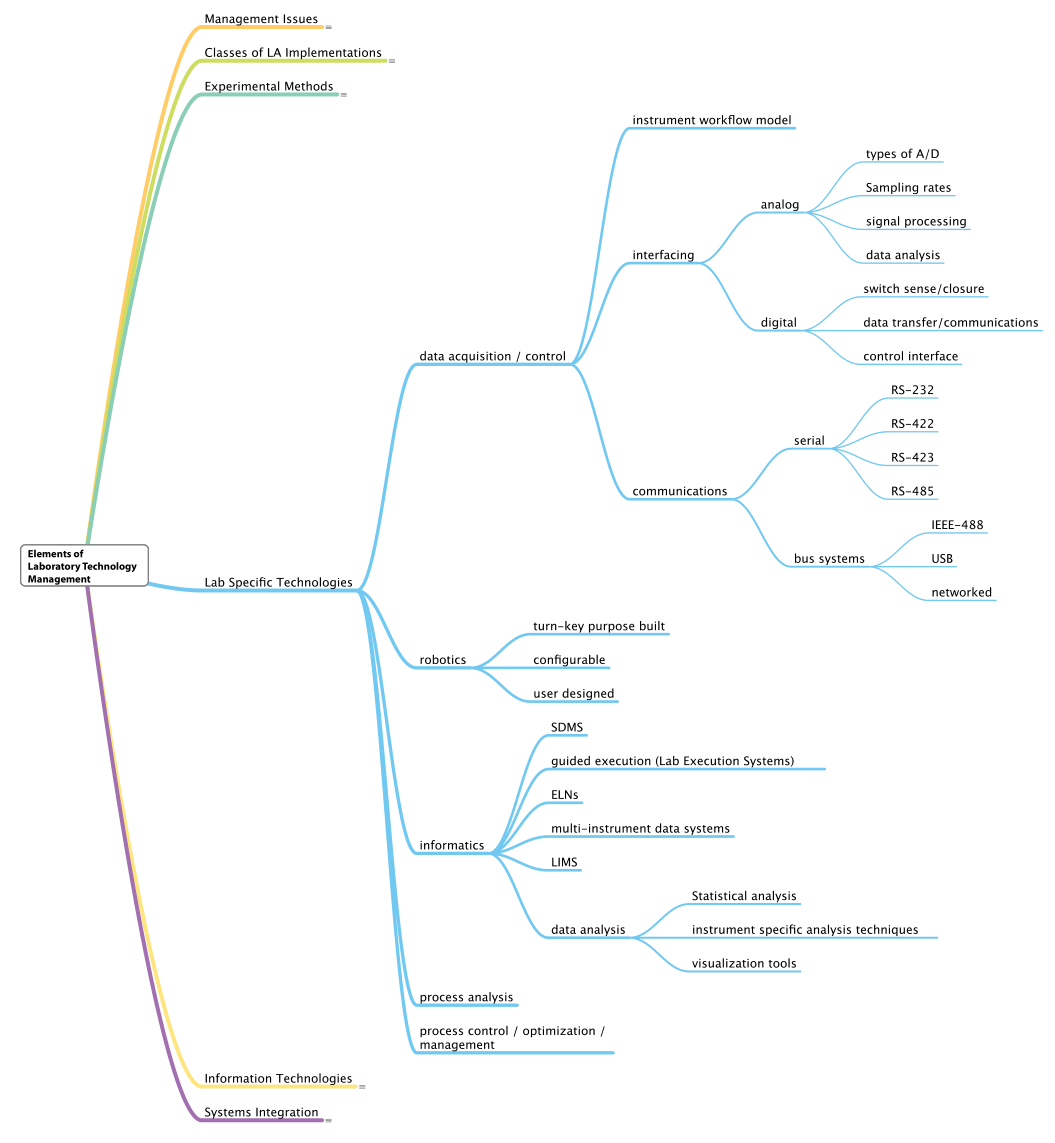
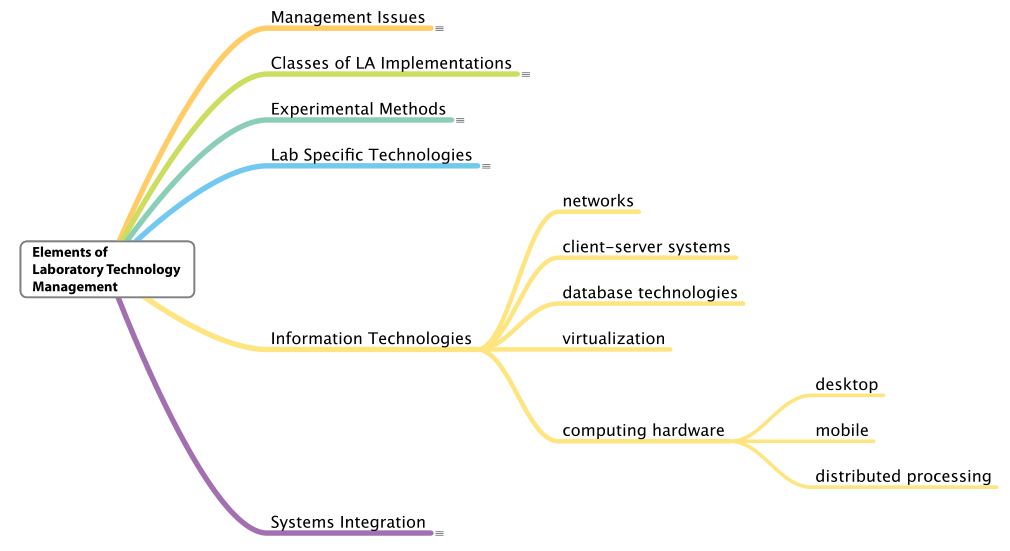
Lab-specific technologies and information technology
This section quickly covers the next two elements of laboratory technology management at the same time, as many aspects of these elements have already been covered elsewhere. See Figure 10 and Figure 11 for a deeper dive into these two elements.[h]
|
|
There is one point that needs to be made with respect to information technologies. While lab managers do not need to understand the implementation details of those technologies, they do need to be aware of the potential they offer in the development of a structure for laboratory automation implementations. Management is responsible for lab automation planning, including choosing the best technologies; in other words, management must manage the “big picture” of how technologies are used to meet their lab's purpose.
In particular, managers should pay close attention to the role of client-server systems and virtualization, since they offer design alternatives that impact the choice of products and the options for managing technology. This is one area where good relationships with IT departments are essential. We’ll be addressing these and other information technologies in more detail in other publications.
Systems integration
Footnotes
- ↑ The Spectronic 20 was developed by Bausch & Lomb in 1954 and is currently owned and marketed in updated versions by ThermoFisher.
- ↑ Mr. Zenie often introduced robotics courses at Zymark with that statement.
- ↑ See ASTM E1578 - 06.
- ↑ This is not a recommended method.
- ↑ This topic will be given light treatment in this work, but will be covered in more detail elsewhere.
- ↑ LabVIEW is a product of National Instruments; similar products are available from other vendors.
- ↑ Having a data system connected to, or in control of, a process is not the same as full automation. For example, there are automated Karl Fisher systems (for water analysis), but they only address titration activities and not sample preparation. A vendor can only take things so far in commercial products unless labs describe a larger role for automation, one that will vary by application. The point is we need a formalized description of that larger context.
- ↑ For more information about lab-specific technologies, please refer to Computerized Systems in the Modern Laboratory: A Practical Guide.
About the author
Initially educated as a chemist, author Joe Liscouski (joe dot liscouski at gmail dot com) is an experienced laboratory automation/computing professional with over forty years of experience in the field, including the design and development of automation systems (both custom and commercial systems), LIMS, robotics and data interchange standards. He also consults on the use of computing in laboratory work. He has held symposia on validation and presented technical material and short courses on laboratory automation and computing in the U.S., Europe, and Japan. He has worked/consulted in pharmaceutical, biotech, polymer, medical, and government laboratories. His current work centers on working with companies to establish planning programs for lab systems, developing effective support groups, and helping people with the application of automation and information technologies in research and quality control environments.
References
- ↑ "Laboratory automation". Wikipedia. Archived from the original on 05 December 2014. https://en.wikipedia.org/w/index.php?title=Laboratory_automation&oldid=636846823.
- ↑ McDowall, R.D. (1993). "A Matrix for the Development of a Strategic Laboratory Information Management System". Analytical Chemistry 65 (20): 896A–901A. doi:10.1021/ac00068a725.