Difference between revisions of "P-Cymene"
Shawndouglas (talk | contribs) (Created as needed) |
Shawndouglas (talk | contribs) m (Text replacement - "CannabisQAwiki" to "LIMSwiki") |
||
| (One intermediate revision by the same user not shown) | |||
| Line 2: | Line 2: | ||
{{wikipedia::P-Cymene}} | {{wikipedia::P-Cymene}} | ||
==Notes== | ==Notes== | ||
This article is a direct transclusion of [https://en.wikipedia.org/wiki/P-Cymene the Wikipedia article] and therefore may not meet the same editing standards as | This article is a direct transclusion of [https://en.wikipedia.org/wiki/P-Cymene the Wikipedia article] and therefore may not meet the same editing standards as LIMSwiki. | ||
<!---Place all category tags here--> | <!---Place all category tags here--> | ||
Latest revision as of 22:32, 28 February 2024
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1-Methyl-4-(propan-2-yl)benzene[1] | |||
| Other names
para-Cymene
4-Isopropyltoluene 4-Methylcumene Paracymene | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1903377 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.542 | ||
| EC Number |
| ||
| 305912 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2046 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C10H14 | |||
| Molar mass | 134.222 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 0.857 g/cm3 | ||
| Melting point | −68 °C (−90 °F; 205 K) | ||
| Boiling point | 177 °C (351 °F; 450 K) | ||
| 23.4 mg/L | |||
| −1.028×10−4 cm3/mol | |||
Refractive index (nD)
|
1.4908 (at 20 °C) [2] | ||
| Hazards | |||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H226, H304, H411 | |||
| P210, P233, P240, P241, P242, P243, P273, P280, P301+P310, P303+P361+P353, P331, P370+P378, P391, P403+P235, P405, P501 | |||
| Flash point | 47 °C (117 °F; 320 K) | ||
| 435 °C (815 °F; 708 K) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
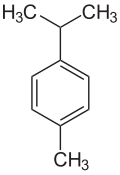
p-Cymene is a naturally occurring aromatic organic compound. It is classified as an alkylbenzene related to monocyclic monoterpenes.[3] Its structure consists of a benzene ring para-substituted with a methyl group and an isopropyl group. p-Cymene is insoluble in water, but miscible with organic solvents.
Isomers and production
In addition to p-cymene, two less common geometric isomers are o-cymene, in which the alkyl groups are ortho-substituted, and m-cymene, in which they are meta-substituted. p-Cymene is the only natural isomer, as expected from the terpene rule. All three isomers form the group of cymenes.
Cymene is also produced by alkylation of toluene with propene.[4]
Related compounds
It is a constituent of a number of essential oils, most commonly the oil of cumin and thyme. Significant amounts are formed in sulfite pulping process from the wood terpenes.
p-Cymene is a common ligand for ruthenium. The parent compound is [(η6-cymene)RuCl2]2. This half-sandwich compound is prepared by the reaction of ruthenium trichloride with the terpene α-phellandrene. The osmium complex is also known.[5]
Hydrogenation gives the saturated derivative p-menthane.
References
- ^ Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. pp. 139, 597. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ Pabst, Florian; Blochowicz, Thomas (December 2022). "On the intensity of light scattered by molecular liquids - Comparison of experiment and quantum chemical calculations". The Journal of Chemical Physics. 157 (24): 244501. Bibcode:2022JChPh.157x4501P. doi:10.1063/5.0133511. PMID 36586992.
- ^ Balahbib, Abdelaali; El Omari, Nasreddine; Hachlafi, Naoufal EL.; Lakhdar, Fatima; El Menyiy, Naoual; Salhi, Najoua; Mrabti, Hanae Naceiri; Bakrim, Saad; Zengin, Gokhan; Bouyahya, Abdelhakim (2021-07-01). "Health beneficial and pharmacological properties of p-cymene". Food and Chemical Toxicology. 153 112259. doi:10.1016/j.fct.2021.112259. ISSN 0278-6915. PMID 33984423.
- ^ Vora, Bipin V.; Kocal, Joseph A.; Barger, Paul T.; Schmidt, Robert J.; Johnson, James A. (2003). "Alkylation". Kirk-Othmer Encyclopedia of Chemical Technology. doi:10.1002/0471238961.0112112508011313.a01.pub2. ISBN 0471238961.
- ^ Bennett, M. A.; Huang, T.-N.; Matheson, T. W.; Smith, A. K. (1982). "16. (η6-Hexamethylbenzene)Ruthenium Complexes". Inorganic Syntheses. Vol. 21. pp. 74–78. doi:10.1002/9780470132524.ch16. ISBN 9780470132524.
Notes
This article is a direct transclusion of the Wikipedia article and therefore may not meet the same editing standards as LIMSwiki.